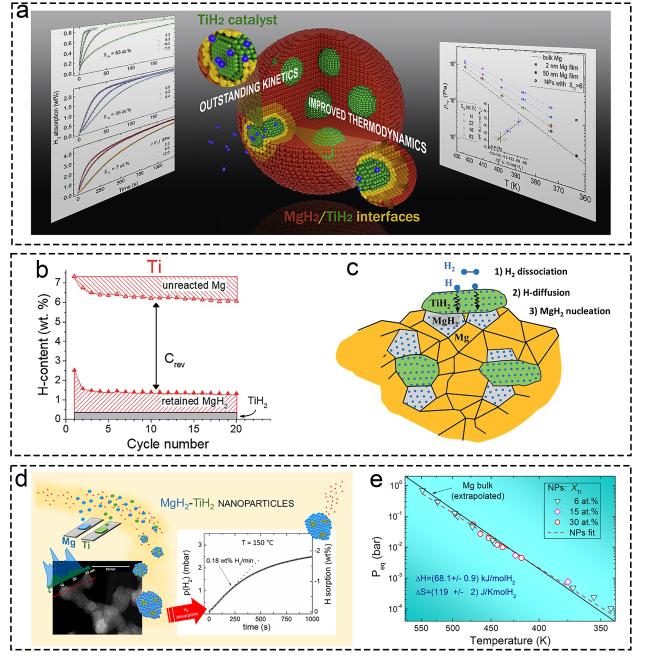
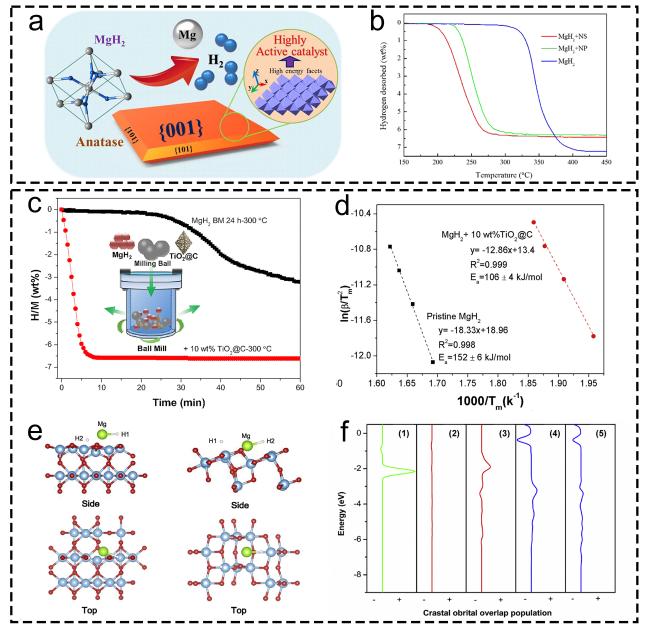
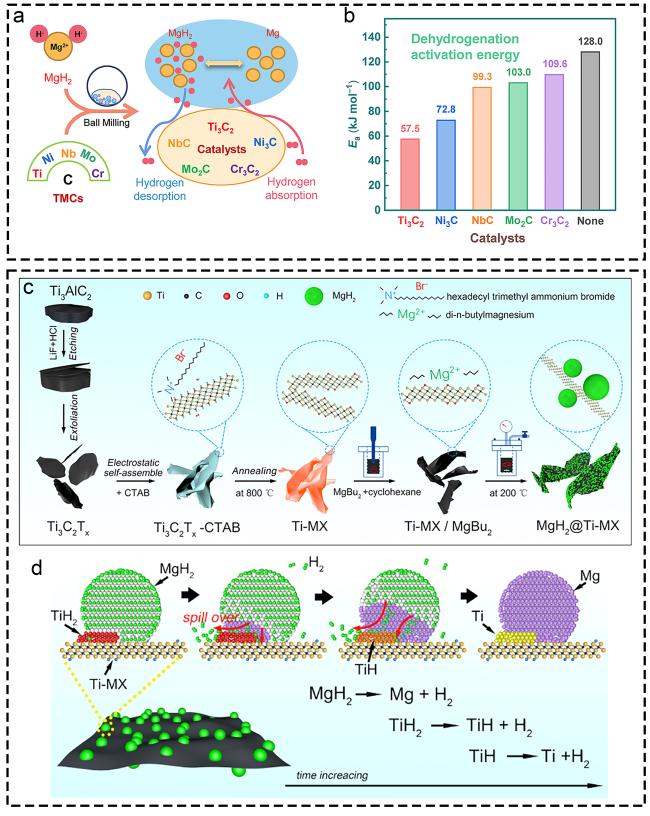
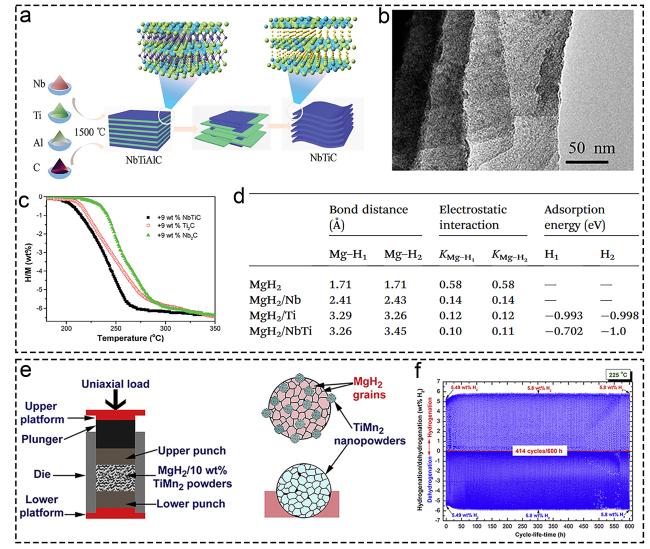
1. Introduction
1.1 Necessary and significance of developing hydrogen storage material
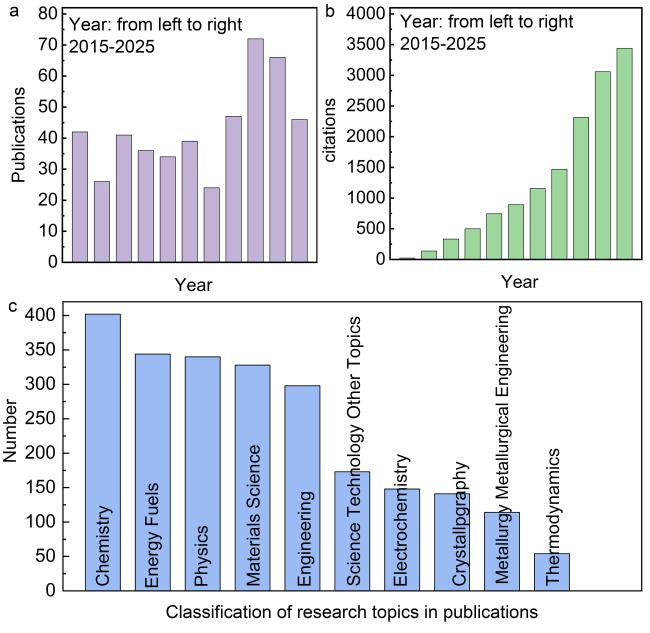
Fig. 1 (a, b) Amounts of publications and citations, (c) corresponding classification of research topics in publications using “Ti-based materials OR Ti materials OR Titanium-based composites”, “Magnesium hydrogen storage” as keywords since 2015. |
1.2 Overall research progress and basic content
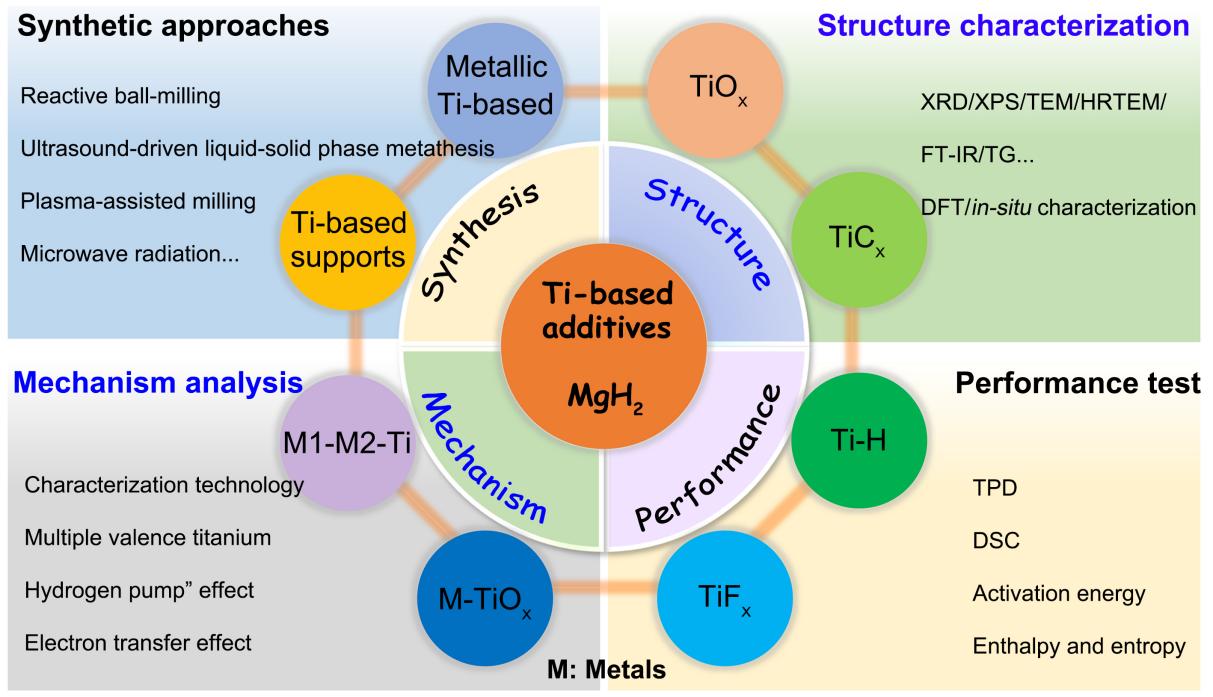
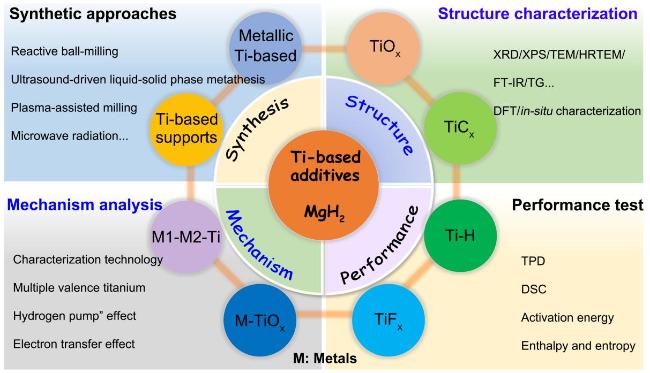
Fig. 2 Schematic illustration of Ti-based additives on hydrogen storage of MgH2. |
1.3 Features and significance of this review
2. Advances in mechanism investigation
2.1 Technologies in analyzing mechanism
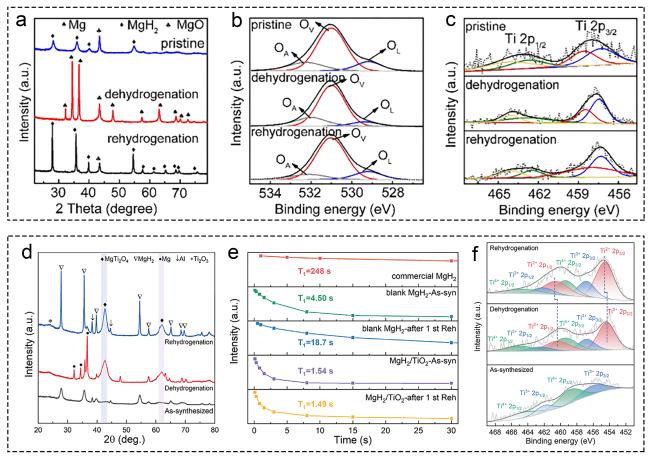
Fig. 3 Mechanism characterization and technology of Ti-based supports. (a-c) MgH2+K2Ti8O17. Reproduced with permission [91]. Copyright 2021, Elsevier. (d-f) MgH2/TiO2. Reproduced with permission [93]. Copyright 2022, Springer. |
2.2 Reaction mechanism
2.2.1 Multiple valence titanium mechanism
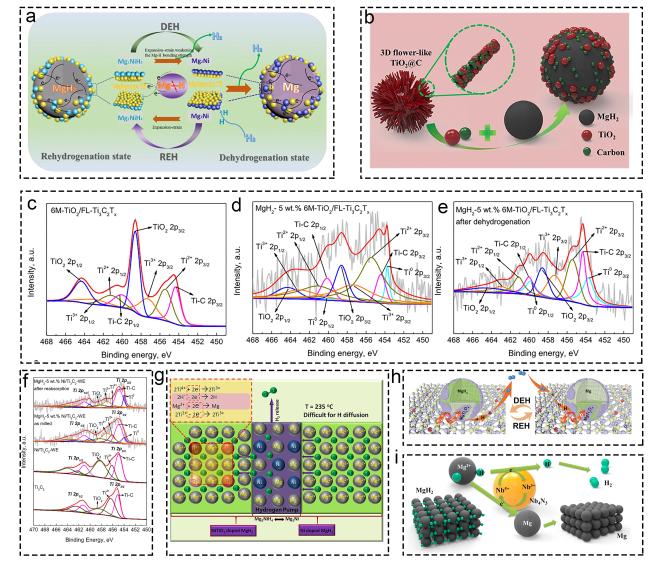
Fig. 4 Investigations on multiple valence titanium mechanism: (a) MgH2-Ni/TiO2. Reproduced with permission [95]. Copyright 2019, Elsevier. (b) MgH2+fl-TiO2@C. Reproduced with permission [96]. Copyright 2019, Elsevier. (c-e) XPS characterization of various materials before and after dehydrogenation. Reproduced with permission [97]. Copyright 2022, Elsevier. (f) Ti3C2-based MgH2 at different states. Reproduced with permission [98]. Copyright 2021, Elsevier. (g) Ti and Ni in MgH2 hydrogen storage system. Reproduced with permission [99]. Copyright 2018, American Chemical Society. (h) MgH2/TiO2. Reproduced with permission [93]. Copyright 2022, Springer. (i) NbN doped MgH2. Reproduced with permission [100]. Copyright 2020, Elsevier. |
2.2.2 “Hydrogen pump” effect mechanism
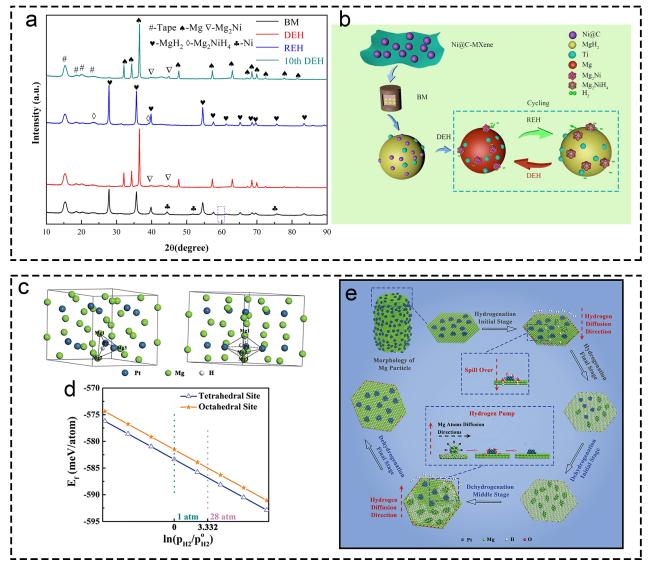
Fig. 5 Investigations on “Hydrogen pump” effect mechanism. (a, b) MgH2+10 wt.% Ni@C-MXene. Reproduced with permission [101]. Copyright 2020, Elsevier. (c-e) Mg@Pt. Reproduced with permission [102]. Copyright 2019, Royal Society of Chemistry. |
2.2.3 Electron transfer effect mechanism
Table 1. The comparison of different reaction mechanisms. |
| Mechanism | Key effect | Application scope |
|---|---|---|
| Multiple valence titanium mechanism | Valence state changes of Ti³⁺/Ti⁴⁺ to catalyze the dissociation of H2 and transport of H⁻ | Ti compounds or precursors |
| “Hydrogen pump” effect | Interface efficiently catalyzes the dissociation/recombination of H2 | Nanoscale metal Ti or in-situ TiHₓ system |
| Electron transfer effect | Weakening strength of Mg-H bond through charge rearrangement | Highly dispersed Ti or forms alloy/solid solution systems |
2.2.4 In-depth analysis of different mechanisms
2.3 Theoretical research of Ti-based additives on hydrogen storage
2.3.1 Purified DFT calculation
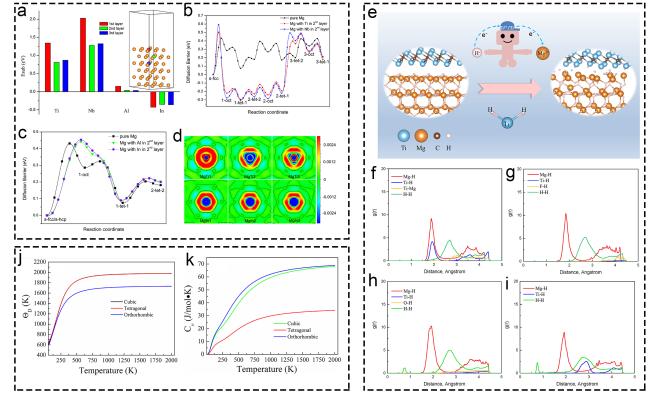
Fig. 6 Theoretical research of Ti-based additives on hydrogen storage based on pure DFT calculation. (a-d) The energy changes of Ti, Nb, Al, and In. Reproduced with permission [109]. Copyright 2017, Elsevier. (e-i) Mg-H, Ti-H and Ti-Mg bond of various structures. Reproduced with permission [111]. Copyright 2021, Elsevier. (j-k) Thermodynamic properties of TiH2 in Debye temperature and heat capacity. Reproduced with permission [112]. Copyright 2020, Wiley. |
2.3.2 Experimental verification guided by DFT calculation
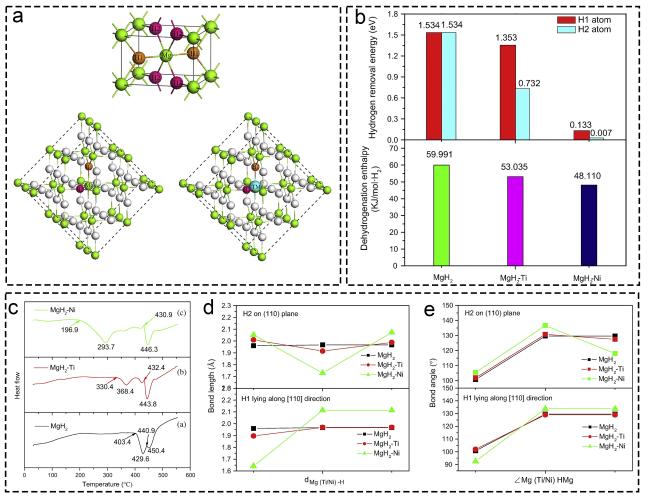
Fig. 7 Theoretical research of Ti-based additives on hydrogen storage based on experimental verification guided by DFT calculation. (a) MgH2 unit cell, Mg16H32 and Mg15TMH32 (TM = Ti or Ni). (b) Calculated dehydrogenation enthalpies and hydrogen removal energies of MgH2-based system. (c) DSC (differential scanning calorimetry) curves of MgH2-based materials. (d, e) Comparison of interatomic bond lengths and bond angles in MgH2-based system. Reproduced with permission [117]. Copyright 2015, Elsevier. |
3. Design strategies of MgH2-based additives
3.1 Reactive ball-milling
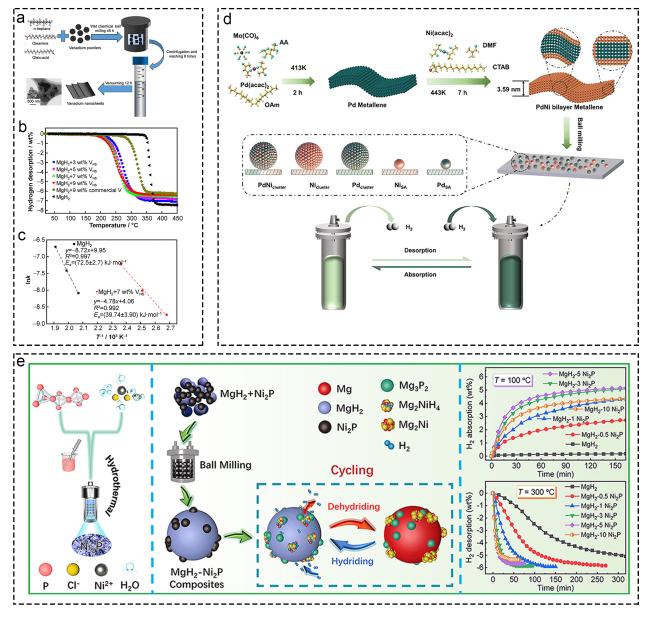
Fig. 8 Corresponding information and presentation of various catalysts on hydrogen storage of MgH2 and MgH2-based composites. (a-c) Vanadium nanosheets. Reproduced with permission [137]. Copyright 2021, Nonferrous Metals Society of China. (d) MgH2-PdNi, Reproduced with permission [138]. Copyright 2023, Wiley. (e) MgH2-Ni2P. Reproduced with permission [140]. Copyright 2022, Elsevier. |
3.2 Plasma-assisted milling
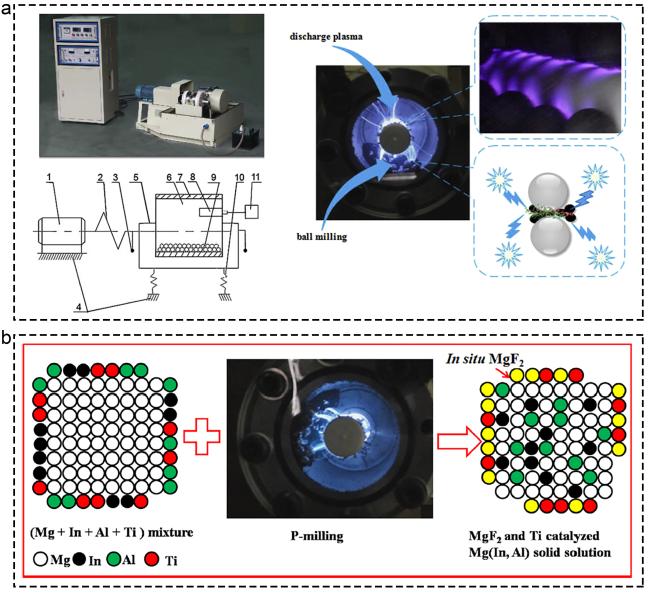
Fig. 9 Corresponding information and presentation of various catalysts on hydrogen storage of MgH2 and MgH2-based composites. (a) P-milling. Reproduced with permission [143]. Copyright 2017, Elsevier. (b) Mg85In5Al5Ti5 alloy by P-milling. Reproduced with permission [146]. Copyright 2015, Elsevier. |
3.3 Ultrasound-dri4.2.1.4 ven liquid-solid phase metathesis
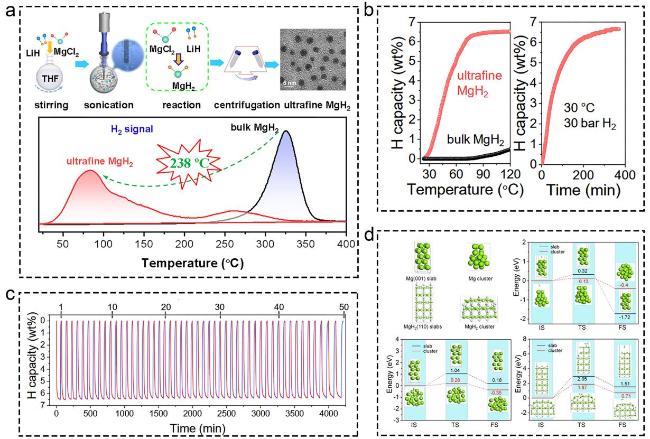
Fig. 10 (a-d) The corresponding information and presentation of ultrafine MgH2 catalysts on hydrogen storage of MgH2 and MgH2-based composites. Reproduced with permission [148]. Copyright 2020, Royal Society of Chemistry. |
3.4 Single atom incorporating
3.5 Other design strategies
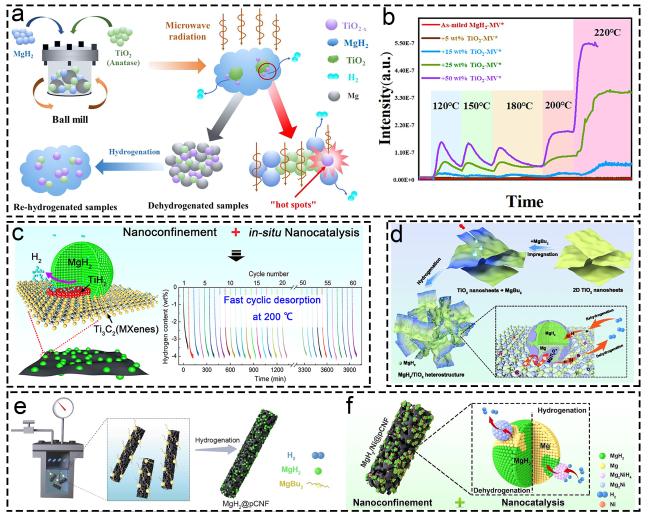
Fig. 11 Corresponding information and presentation of different catalysts on hydrogen storage of MgH2 and MgH2-based composites. (a, b) MgH2-TiO2. Reproduced with permission [190]. Copyright 2022, Elsevier. (c) MgH2@Ti-MX. Reproduced with permission [40]. Copyright 2021, American Chemical Society. (d) MgH2/TiO2. Reproduced with permission [93]. Copyright 2022, Springer. (e, f) MgH2@pCNF. Reproduced with permission [192]. Copyright 2022, Elsevier. |
Table 2. The comparison of different reaction strategies. |
| Methods | Advantages | Disadvantages | Cost | Applicable scenarios |
|---|---|---|---|---|
| Ball milling | 1. Easy to operate 2. Low cost 3. Wide applicability 4. Industrialization potential | 1. Less efficient 2. Introduce pollution 3. Limited particle refinement | Low | 1. Basic research 2. Preliminary exploration |
| Plasma-assisted milling | 1. Extremely efficient 2. Excellent dispersion 3. Clean and activate surface | 1. Complex equipment 2. Ultra-high cost 3. Complex process | High | 1. High-level research 2. Maximizing catalysis 3. Exploring extreme performance |
| Ultrasonication | 1. Achieved true nanoscale dispersion 2. Avoid unnecessary phase changes or reactions | 1. Solvents must be used 2. Extremely low yields 3. Not suitable for mass production | Medium | Basic mechanism research |
4. Universal research procedures in hydrogen storage system
4.1 Properties of hydrogen adsorption and desorption
4.2 Evaluation of reaction kinetics and thermodynamics
4.2.1 Johanson-Mehl-AvramiKolmogorov model
4.2.2 Kissinger’s method
4.2.3 Reaction enthalpy and entropy
5. Investigation on design of Ti-based additives
5.1 Designs aimed at providing active sites
Fig. 12 Corresponding characterization from the morphology, performance and mechanism characterization of Ti-based materials. (a) Reproduced with permission [249]. Copyright 2020, Elsevier. (b, c) Reproduced with permission [251]. Copyright 2019, Royal Society of Chemistry. (d, e) 3D Ti3C2 MXene. Reproduced with permission [252]. Copyright 2017, American Chemical Society. |
5.2 Designs aimed at promoting electron transfer
Fig. 13 Corresponding information and presentation of different catalysts on hydrogen storage of MgH2 and MgH2-based composites. (a, b) TiO2 NS with exposed {001} facets. Reproduced with permission [72]. Copyright 2019, Royal Society of Chemistry. (c-f) TiO2@C. Reproduced with permission [274]. Copyright 2018, Elsevier. |
5.3 Designs aimed at creating synergistic effects
Fig. 14 Morphology, performance and mechanism characterization of TiCx-based materials. (a, b) Some transitional metal carbides. Reproduced with permission [292]. Copyright 2021, Elsevier. (c, d) 3D Ti3C2 MXene. Reproduced with permission [40]. Copyright 2021, American Chemical Society. |
Fig. 15 The corresponding characterization from the morphology, performance and mechanism characterization of polymetallic titanium compounds. (a-d) NbTi cluster. Reproduced with permission [299]. Copyright 2019, Royal Society of Chemistry. (e, f) TiMn2 compound. Reproduced with permission [300]. Copyright 2019, Royal Society of Chemistry. |
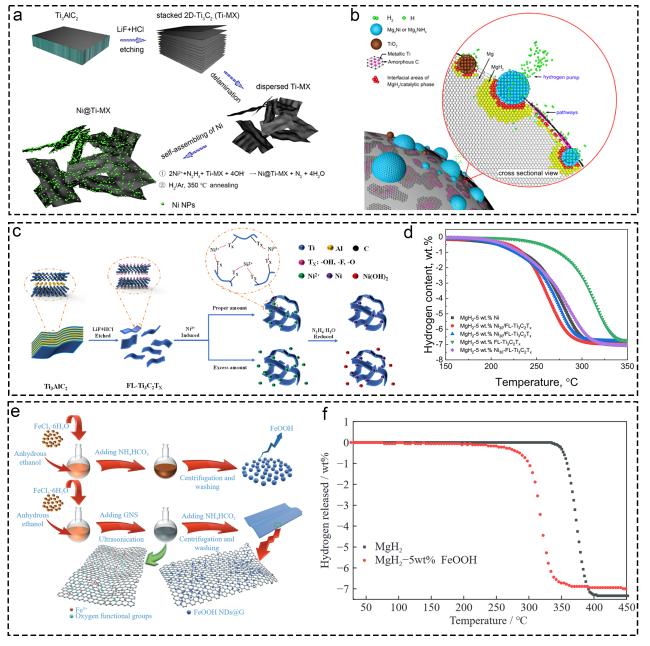
Fig. 16 The corresponding characterization from the morphology, performance and mechanism characterization of materials supported on Ti-based supports. (a, b) Ti3C2 MXene-based catalyst (Ni@Ti-MX). Reproduced with permission [315]. Copyright 2020, American Chemical Society. (c, d) few-layer Ti3C2Tx (FL-Ti3C2Tx) supporting highly dispersed nano-Ni particles. Reproduced with permission [316]. Copyright 2020, American Chemical Society. (e, f) graphene nanosheet-supported FeOOH nanodots. Reproduced with permission [318]. Copyright 2022, Springer. |
5.4 Designs aimed at destabilizing thermodynamics
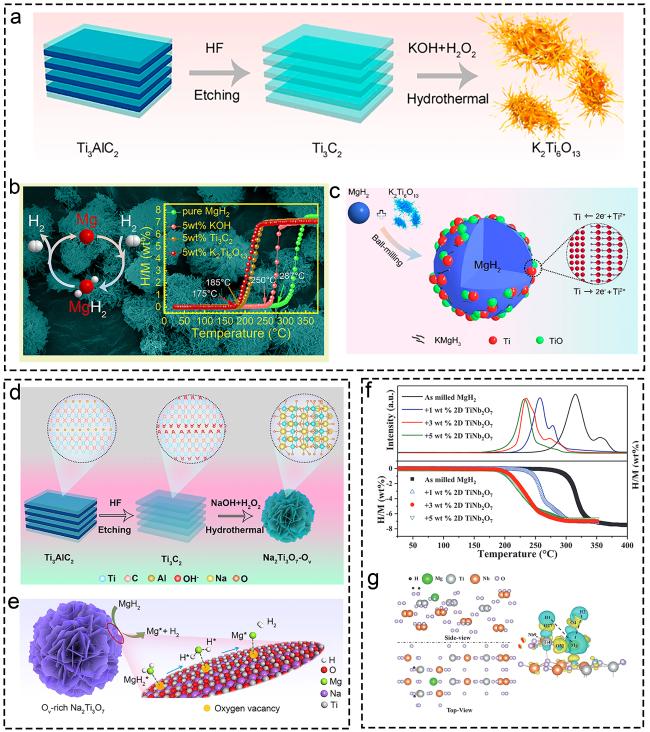
Fig. 17 Corresponding characterization from the morphology, performance and mechanism characterization of Ti-based materials. (a-c) K2Ti6O13. Reproduced with permission [333]. Copyright 2020, American Chemical Society. (d, e) Oxygen Vacancies modified Na2Ti3O7. Reproduced with permission [90]. Copyright 2022, American Chemical Society. (f, g) TiNb2O7. Reproduced with permission [335]. Copyright 2022, Wiley. |
Table 3. The performance comparison of different Ti-based materials. |
| Number | Materials | Onset dehydrogenation temperature (°C) | Release capacity (wt.%) | Activation energy (kJ/mol) | Ref. |
|---|---|---|---|---|---|
| 1 | MgH2-12Ti | 300 | 6.39 | - | [239] |
| 2 | 85MgH2+15Ti | 320 | 5.51 | - | [240] |
| 3 | Mg@Ti@Ni | 400 | 6.27 | 63.7 | [242] |
| 4 | Mg-Ti NPs | 280 | 5.3 | 68 | [243] |
| 5 | AlCl3/Ti | 400 | 6.0 | 113.7 | [245] |
| 6 | Ti-Mn-Cr | 362 | - | - | [247] |
| 7 | Mg-Ti-H | 150 | 5.0 | - | [249] |
| 8 | Ti/MgH2 | - | - | 89.4 | [250] |
| 9 | MgH2-TiH2 | - | 7.3 | - | [251] |
| 10 | MgH2-TiH2 NCs | 300 | 4.9 | - | [253] |
| 11 | MgH2-TiO2 | 200 | 6.29 | 75.3 | [263] |
| 12 | MgH2+ 5wt% TiO2 NS | 180.5 | 6.0 | 67.6 | [72] |
| 13 | MgH2-TiO2 | 350 | 5.5 | - | [264] |
| 14 | MgH2-DOM TiO2 | 300 | 5.75 | 72.8 | [265] |
| 15 | MgH2/5wt% TiO2 | 307 | - | 110.9 | [266] |
| 16 | K-TiO2−x | 194 | 6.0 | 69.1 | [268] |
| 17 | MgH2-10wt% TiO2@C | 205 | 6.5 | 106 | [274] |
| 18 | MgH2-TiO2@C | 199.2 | 6.0 | 67.10 | [96] |
| 19 | MgH2-TiO2 SCNPs/AC | 163.5 | 6.5 | 69.2 | [275] |
| 20 | MgH2-Ni/TiO2 | 232 | 6.5 | 43.7 | [95] |
| 21 | MgH2-Ti3AlCN | 205 | 6.6 | 94.2 | [282] |
| 22 | Mg+7wt% MAX | 236 | 5.6 | 60.9 | [283] |
| 23 | MgH2-7wt% Ti3AlC2 | 205 | 6.9 | 104.7 | [284] |
| 24 | MgH2-Ti3C2 | 205 | 4.6 | 57.5 | [292] |
| 25 | MgH2-5 wt%Ti2C | - | - | 157.9 | [293] |
| 26 | 60MgH2@Ti-MX | 140 | 6.4 | - | [40] |
| 27 | 5wt% Ti3C2-MgH2 | 300 | 6.2 | - | [294] |
| 28 | MgH2/5TiC/5Fe-12Cr | 275 | 5.5 | 97.74 | [295] |
| 29 | Mg96(TiC)4@C | 400 | 6.5 | 56.5 | [296] |
| 30 | Mg-20wt % Ti0.16Cr0.24V0.6 | 224 | - | 76.32 | [297] |
| 31 | MgH2-20wt% Ti0.35Cr0.45V0.2 | 256 | 5.30 | 86.43 | [298] |
| 32 | MgH2-9wt% NbTiC | 195 | 6.8 | 80.0 | [299] |
| 33 | MgH2/10wt% TiMn2 | 225 | 5.1 | 82.9 | [300] |
| 34 | MgH2+10wt%-TiFe | 175 | 6.5 | 60.7 | [135] |
| 35 | MgH2+Ni@Ti-MX | 250 | 5.2 | 73.0 | [315] |
| 36 | MgH2-5wt% Ni30-FLTi3C2Tx | 250 | 5.83 | 106.92 | [316] |
| 37 | MgH2-5wt% Ti3C2/TiO2(A)-C | 125 | 4.0 | 77.69 | [317] |
| 38 | MgH2-10wt% FeOOH NDs@G | 325 | 6.3 | 125.03 | [318] |
| 39 | MgH2+5wt% K2Ti6O13 | 175 | 6.7 | 105.67 | [333] |
| 40 | MgH2-Na2Ti3O7 NTs | 233.5 | 6.5 | 70.43 | [334] |
| 41 | MgH2-TiNb2O7 | 178 | 5.7 | 100.4 | [335] |
| 42 | MgH2+20wt% BaTiO3 | 270 | - | 108 | [337] |
| 43 | MgH2-10wt% SrTiO3-Ni | 260 | 6.0 | 98.6 | [338] |
| 44 | MgH2-6wt% NiTiO3 | 235 | 7.4 | 74 | [99] |
| 45 | Mg-NiTiO3/TiO2 | 193.2 | 6.6 | 69.8 | [134] |
| 46 | MgH2-Li2TiO3 | 170 | - | 84 | [339] |
| 47 | MgH2-5wt% PrF3/Ti3C2 | 180 | 7.0 | 78.11 | [92] |
5.5 Conclusion and reflection of Ti-based materials
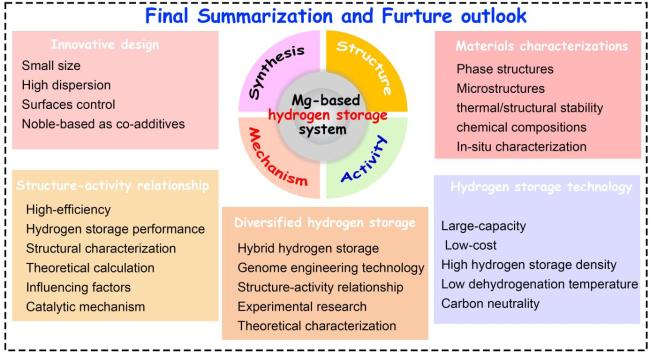
6. Final summary and future outlook
6.1 Innovative design of Ti-based additives
6.2 Characterizations of MgH2 composites in hydrogen storage system
Fig. 18 Final summary and future outlook of Ti-based additives on Mg-based hydrogen storage system. |