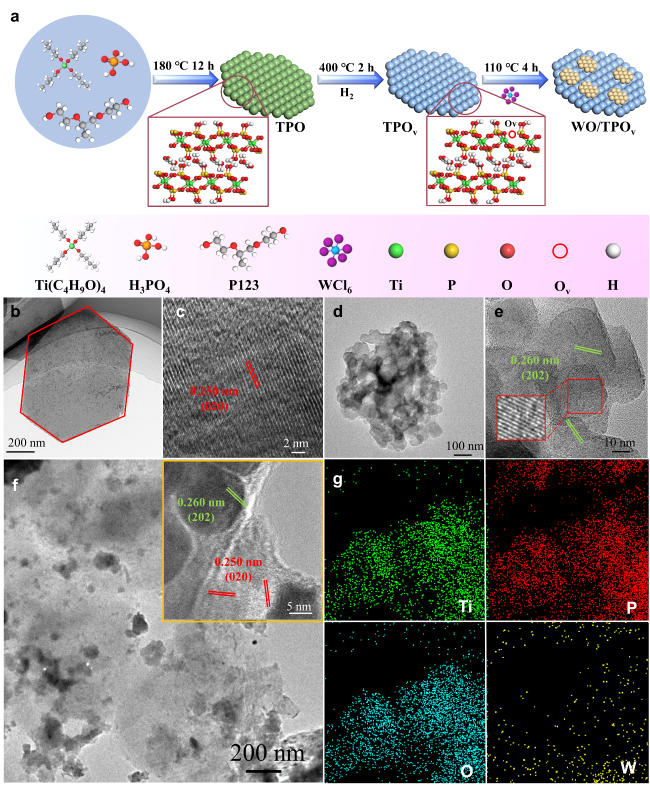
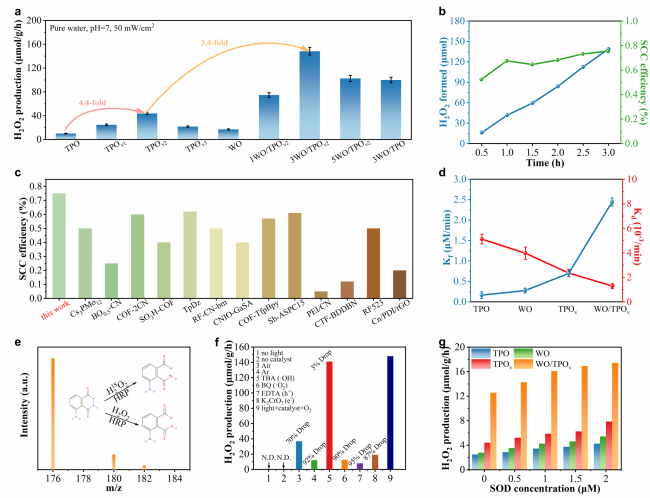
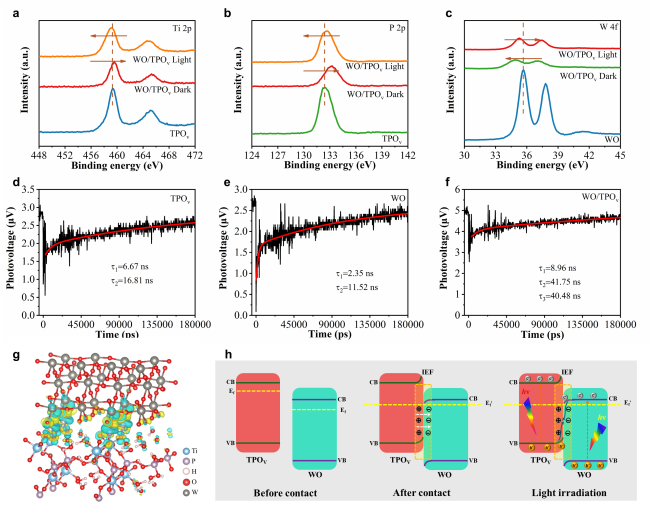
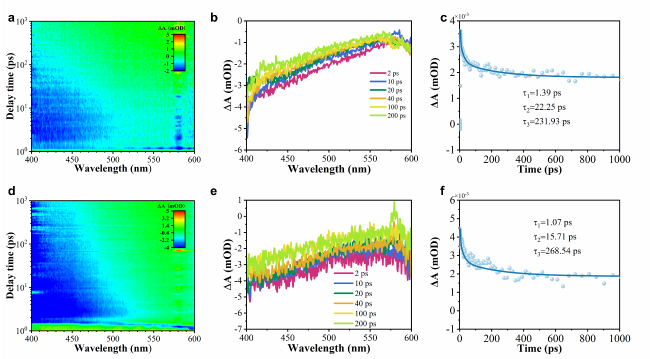
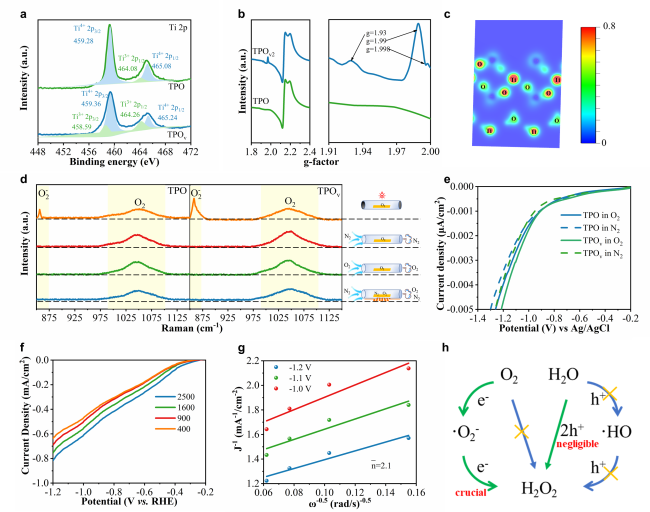
To deeply investigate the impact of the Ti
3+-O
v-P structure on the O
2 activation effect, we first systematically examined its O
2 adsorption capacity. By analyzing the data in Figures S21 and S22, we found that in the TPO
v catalyst, with the extension of O
2 adsorption duration, the characteristic binding energy peak corresponding to adsorbed oxygen in the in-situ O1s XPS spectrum has been significantly higher than the corresponding peak of the TPO catalyst since the initial stage of the experiment, indicating that the TPO
v catalyst possesses a stronger O
2 adsorption activity. Further O
2-TPD experimental results also confirmed this point [
70-
72]. Whether considering chemisorption or physisorption, the O
2 adsorption activity of the TPO
v catalyst is significantly better than that of the TPO material. These findings lay a solid foundation for subsequent O
2 activation capacity testing. Prior to the formal
in-situ Raman spectroscopy measurements, a series of pretreatment operations were conducted, encompassing the elimination of surface contaminants, adsorption of O
2, and purging with N
2 to ensure the reliability of subsequent test results. As illustrated in
Figure 5d, the Raman spectrum reveals that the characteristic Raman peak assigned to adsorbed O
2 remains essentially unchanged for the same material throughout the entire testing period, confirming the absence of free O
2 interference during detection [
73-
75]. Furthermore, the intensity of the adsorbed oxygen-related Raman peak for TPO
v is consistently higher than TPO, which further verifies that the Ti
3+-O
v-P structure enhances the O
2 adsorption capability. Notably, upon introducing light into the monitoring system, TPO
v exhibits a more prominent ·O₂⁻ characteristic peak, indicative of its superior oxygen activation performance. This finding is corroborated from an alternative perspective via atmosphere-controlled electrochemical oxygen reduction measurements (
Figure 5e). Upon switching the purge gas from N
2 to fully O
2-saturated electrolyte, the potential required to activate O
2 on TPO
v material dropped markedly below that observed for TPO material. In addition, the SS-SPS spectra with different atmospheres (Figure S23) and DMPO-·O
2- EPR spectrum (Figure S24) also provided evidence supporting this conclusion. From the perspective of electronic structure essence, the projected density of states clearly reveals the decisive regulatory role of oxygen vacancies (O
V) in the catalytic activity of TPO (Fig. S25). For the vacancy-free sample, the density of states near the Fermi level (E
f) is almost vacant with a wide band gap. The Ti 3d and O 2p orbitals are locally confined, failing to provide sufficient e
- to the O
2 π* orbitals, thus oxygen activation is restricted by the electron transfer prohibition. After the introduction of oxygen vacancies, to strictly maintain the overall electrical neutrality of the material, the positive charge defects released by oxygen vacancy formation trigger a local charge self-compensation mechanism. Under the action of this mechanism, Ti
4+ ions adjacent to the oxygen vacancies are selectively occupied by e
- and reduced to Ti
3+, thereby forming a local polaron state with high electron density in the defect region, and the 3d
1 e
- form new energy levels within the band gap. These levels rehybridize with the relaxed O 2p orbitals around the vacancies, generating continuous defect states with high intensity near E
f. Such defect states simultaneously satisfy three electronic criteria for oxygen activation: (1) The density of states crosses E
f, enabling immediate participation of e
- in the reaction; (2) The significant overlap between Ti 3d and O 2p peaks achieves effective coupling with the O₂ π* orbitals; (3) The significant increase in the density of states at E
f, greatly enhancing the electron supply capacity. The experimentally observed enhancement in oxygen activation activity is thereby directly explained at the electronic structure level. In summary, through a series of experiments, we have demonstrated that the Ti
3+-O
v-P structure has significant advantages in O
2 adsorption and activation.