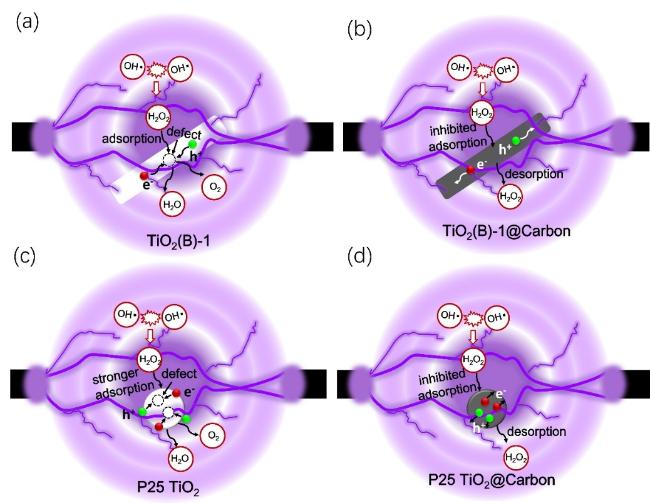
When H
2O
2 does not dissociate immediately from the catalyst surface, electrons migrating to the surface can reduce H
2O
2 to H
2O. Additionally, TiO
2 surfaces form Ti-OOH complexes with H
2O
2, promoting its decomposition under visible light irradiation. To investigate the diffusion behavior of H
2O
2 across different crystalline phases and exposed surfaces, we designed experiments to observe H
2O
2 decomposition under UV light.
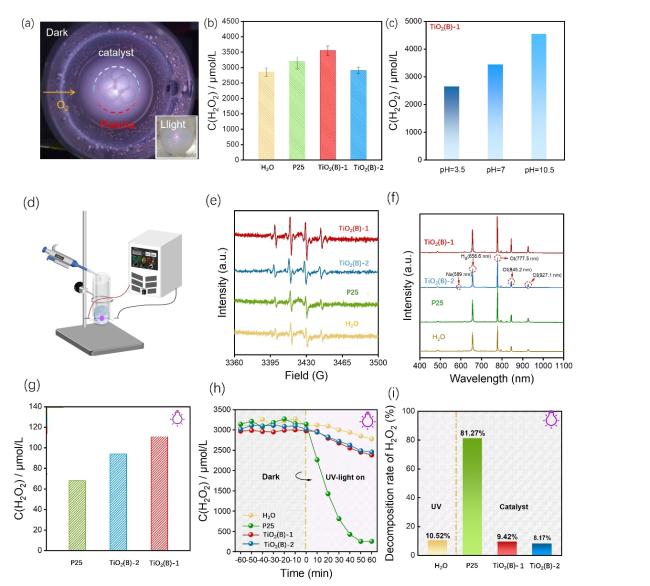
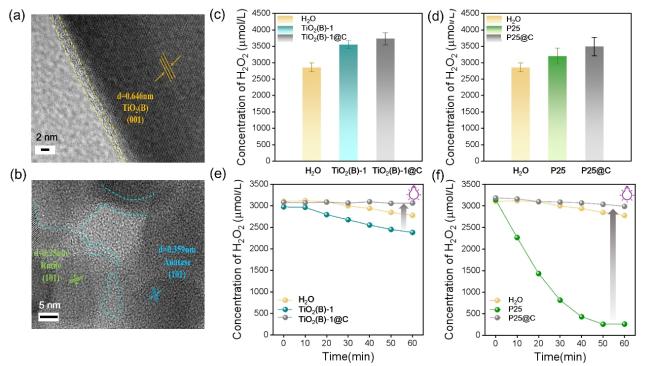
Figure 4h shows the decomposition effect of H
2O
2 with the catalyst under UV irradiation. Upon UV exposure, H
2O
2 decomposed gradually. In pure water, the decomposition rate was 10.52%, while both TiO
2-based catalysts exhibited slow decomposition rates. Notably, P25 showed the highest H
2O
2 decomposition ability, achieving an 81.27% decomposition rate under UV light. These differences are attributed to the exposed (001) crystal plane of bronze TiO
2, which facilitates excellent H
2O
2 desorption, leading to a decrease in the decomposition rate. This can be also supported by the results in the literature
[30]: On one hand, the diffusion coefficient of H
2O
2 on the surface of bronze-phase TiO
2 is significantly higher than that on anatase-phase TiO
2. This indicates that H
2O
2 desorbs more readily from the bronze-phase TiO
2 surface, which is beneficial for suppressing its decomposition. On the other hand, the adsorption heat of H
2O
2 on bronze-phase TiO
2 (11.3 kJ/mol) is notably lower than that on bronze/anatase TiO
2 heterojunctions (19.4 kJ/mol). A higher adsorption heat corresponds to stronger adsorption of H
2O
2 on the TiO
2 surface. Therefore, the lower adsorption heat of H
2O
2 on bronze-phase TiO
2 compared to anatase-phase TiO
2 suggests weaker adsorption of H
2O
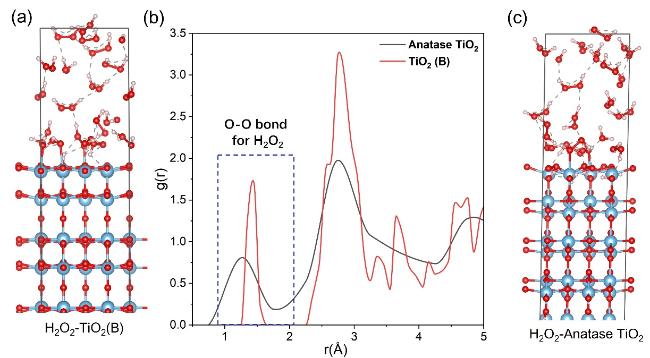
2. Besides, we conducted a radial distribution function analysis of the O-O distance distribution from the AIMD results of H
2O
2 on the TiO
2 surface. The molecular dynamics simulation of H
2O
2-TiO
2(B) and H
2O
2-anatase TiO
2 configuration are depicted in
Figure 5a and c. Figure S9 indicates that their kinetic structural relaxations reach a steady state. The simulation results are displayed in
Figure 5b. The peak around 1.3 Å corresponds to the O-O bond length in H
2O
2. By analyzing the g(r)-r relationships, we can effectively compare the stability of H
2O
2 molecules on different TiO
2 surfaces. Upon comparison, we observe that the proportion of stable H
2O
2 molecules on the TiO
2(B) surface is significantly higher than that on the anatase phase of TiO
2, which is revealed by a higher value of g(r) for TiO
2(B). This indicates that after kinetic relaxation, TiO
2(B) retains more H
2O
2 on its surface, which is more favorable for stabilizing H
2O
2.