1. Introduction
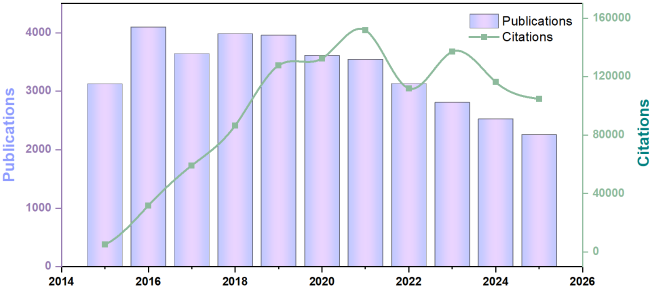
Figure 1 Number of citations and publications of research on hollow-structured materials in Web of Science over time. |
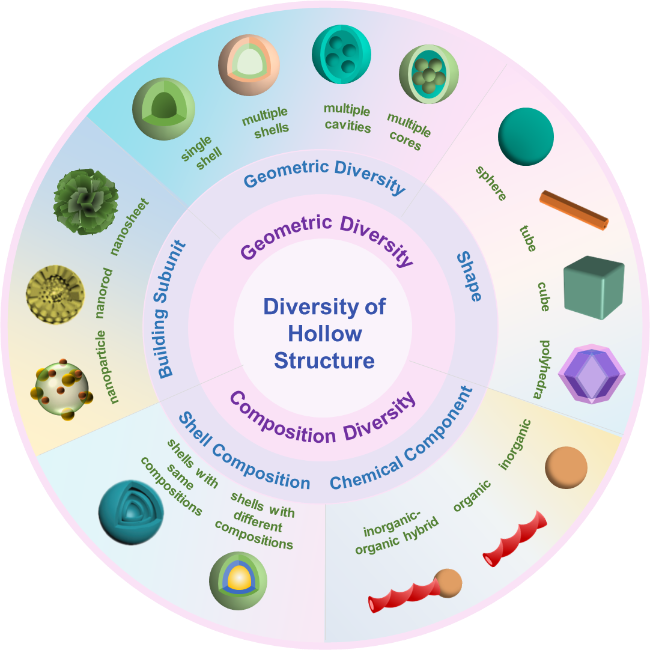
Scheme 2 Diversity of hollow structure. |
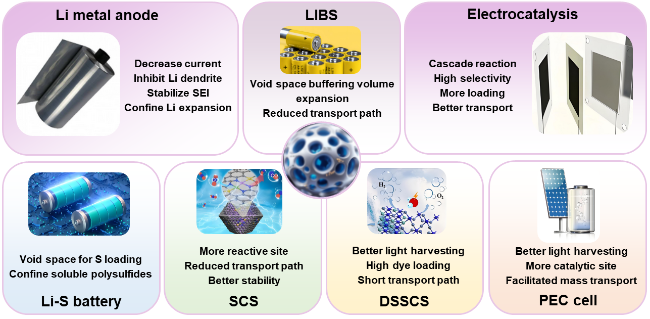
Scheme 1 Advantages of hollow nanostructures for energy storage applications. |
2. Synthesis Methods of Hollow Structures
Table 1 Typical research contents related to hollow-structured materials in Scheme 3. |
| Year | Hollow structural materials type | Synthetic method | Ref. |
|---|---|---|---|
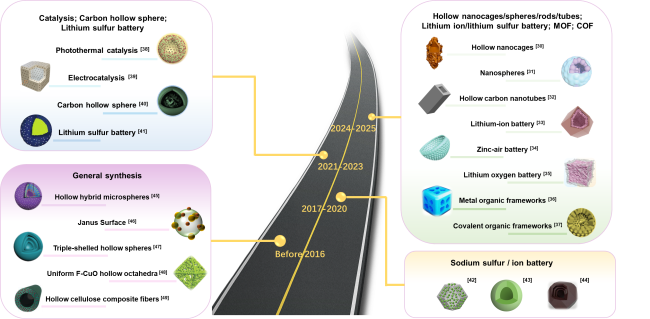
| 2024-2025 | Hollow nanocages | Template-free method | [30] |
| Hollow nanospheres | Template-free method | [31] | |
| Hollow nanotubes | Template-free method | [32] | |
| Hollow core-shell dodecahedra | Hard template method | [33] | |
| Hollow bowl | Hard template method | [34] | |
| Hollow nanocage | Template-free method | [35] | |
| Hollow metal organic frameworks | Template-free method | [36] | |
| Hollow covalent organic frameworks | Soft template method | [37] | |
| 2021-2023 | Hollow core-shell nanoparticles | Hard template method | [38] |
| Hollow nanocubes | Hard template method | [39] | |
| Carbon hollow spheres | Hard template method | [40] | |
| Hollow microspheres | Hard template method | [41] | |
| 2017-2020 | Hollow Janus-Featured | Hard template method | [42] |
| Hollow nanospheres | Hard template method | [43, 44] | |
| Before 2016 | Hollow hybrid microspheres through inward crystallization process | Template-free method | [45] |
| Hollow carbon nitride structure with a Janus Surface | Hard template method | [46] | |
| Triple-shelled hollow spheres | Template-free method | [47] | |
| Uniform F-CuO hollow octahedra | Template-free method | [48] | |
| Hollow materials by fast pyrolysis of cellulose composite fibers | Template-free method | [49] |
Scheme 3 A brief timeline of the development history of research on the synthesis and application of hollow-structured materials. |
2.1 Hard Template Synthesis
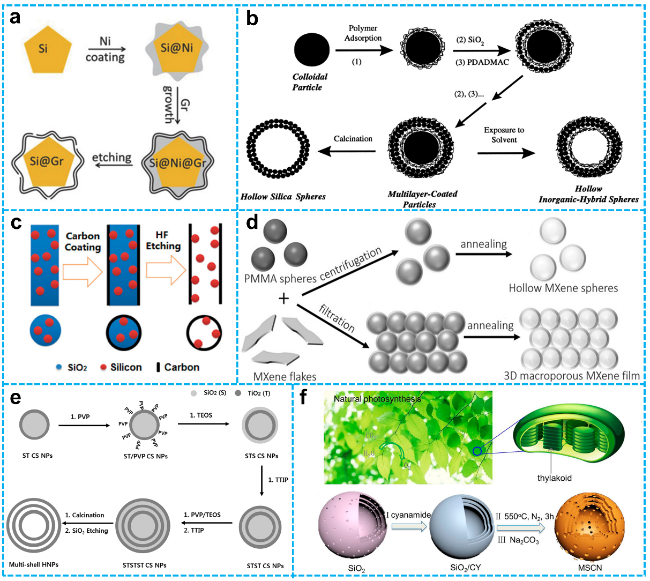
Figure 2 a) Schematic diagram of the synthesis of SiMP@Gr yolk-shell structure [59]. Copyright 2016, Springer Nature. b) Description of synthetic protocols for inorganic and hybrid hollow spheres [58]. Copyright 1998, The American Association for the Advancement of Science. c) Overview of the manufacturing of hollow carbon tubes encapsulated with Si nanoparticles [60]. Copyright 2012, American Chemical Society. d) Diagram illustrating the synthesis of hollow MXene spheres and MXene-Derived 3D macroporous frameworks [61]. Copyright 2017, John Wiley and Sons. e) Schematic illustration for the hard templating synthesis of TiO2 TSHSs [62]. Copyright 2014, John Wiley and Sons. f) Schematic illustration for the hard templating synthesis of MSCN [63]. Copyright 2017, American Chemical Society. |
2.2 Soft Template Synthesis
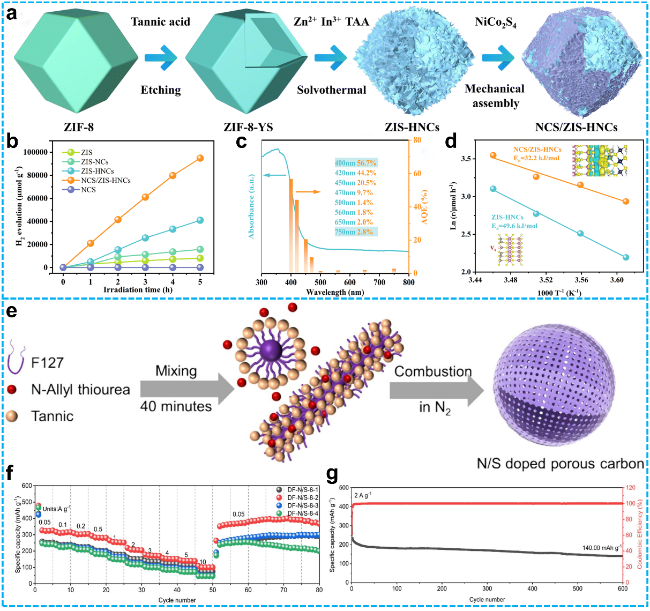
Figure 3 a) Schematic illustration of the fabrication pathway for NCS/ZIS-HNC heterostructures; Photocatalytic hydrogen evolution (PHE) performance of the prepared samples; c) Apparent quantum efficiency (AQE) and diffuse reflectance spectroscopy (DRS) spectral data for NCS/ZIS-HNCs; d) Temperature-dependent PHE rate analysis of ZIS-HNC and NCS/ZIS-HNC materials [52]. Copyright 2023, Royal Society of Chemistry. e) Schematic diagram of the synthetic procedures for DF-N/S-8-2; f) Rate capability at different doping levels; g) Cycling stability tested at a current density of 2 A·g-1 [53]. Copyright 2024, Elsevier. |
2.3 Template-Free Synthesis
2.3.1 Selective Etching
2.3.2 Ostwald Ripening
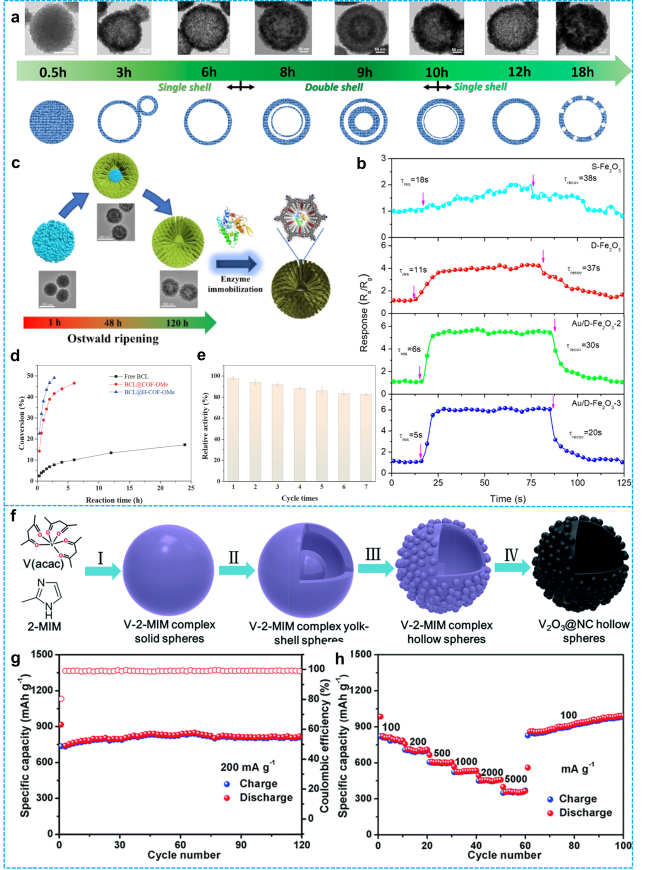
Figure 4 a) Structural evolution of Fe2O3 spheres as a function of reaction duration; b) Dynamic response profiles of four gas sensors to 10 ppm acetone at 200°C [81]. Copyright 2019, American Chemical Society. c) Self-template method for preparing hollow lipase (BCL@H-COF-OMe) via Ostwald ripening mechanism; d) Comparison of catalytic performance of different catalysts for kinetic resolution of l-phenyl ethanol; e) Recycling catalytic performance tests of BCL@H-COF-OMe for kinetic resolution of 1-phenylethanol [55]. Copyright 2022, Springer Nature. f) Schematic representation of the formation process for uniform V2O3@NC hollow spheres; g) Cycling stability at a low current density of 200 mA·g-1; h) Rate capability at current densities of 100, 200, 500, 1000, 2000, 5000, and back to 100 mA·g-1 [82]. Copyright 2018, Royal Society of Chemistry. |
2.3.3 Kirkendall Effect
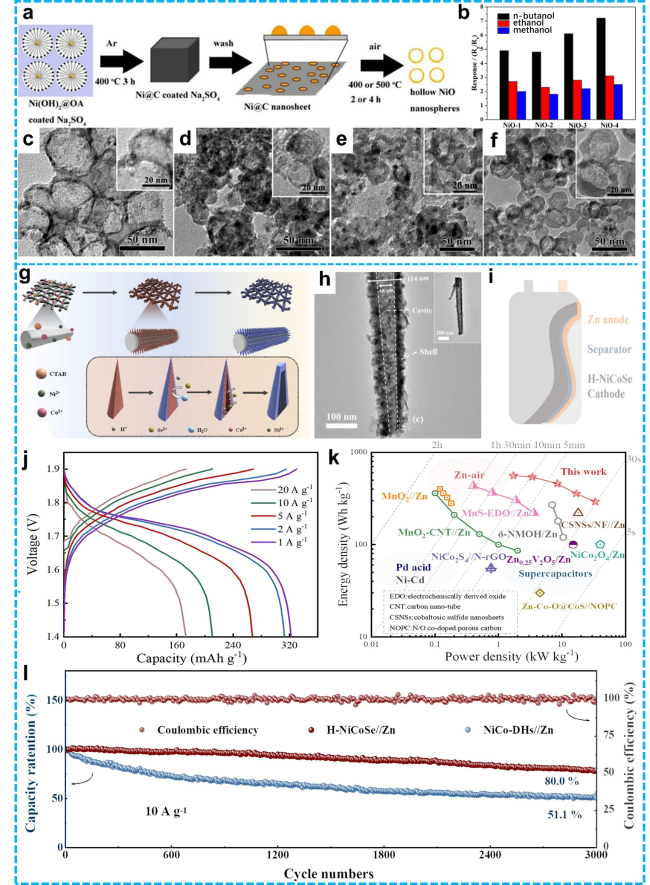
Figure 5 a) Schematic diagram of hollow NiO nanosphere synthesis procedures; b) Selectivity of four NiO-based sensors toward 800 ppm target gases at 240°C; TEM images of (c) NiO-1; (d) NiO-2; (e) NiO-3; (f) NiO-4. The inset images show cross-sectional TEM views of hollow NiO nanospheres [84]. Copyright 2019, Elsevier. g) Diagram depicting the synthesis route of H-NiCoSe@CC nanoarrays; h) TEM image of the prepared H-NiCoSe nanoarrays; i) Schematic representation of a flexible full battery using H-NiCoSe as the cathode and zinc metal as the anode; j) Galvanostatic charge-discharge (GCD) profiles of the H-NiCoSe//Zn battery at various current densities; k) Ragone plots for the H-NiCoSe//Zn battery; l) Cycling performance of the as-assembled H-NiCoSe//Zn and NiCo-DHs//Zn batteries at a current density of 10 A·g-1 [54]. Copyright 2023, Elsevier. |
2.3.4 Ion Exchange
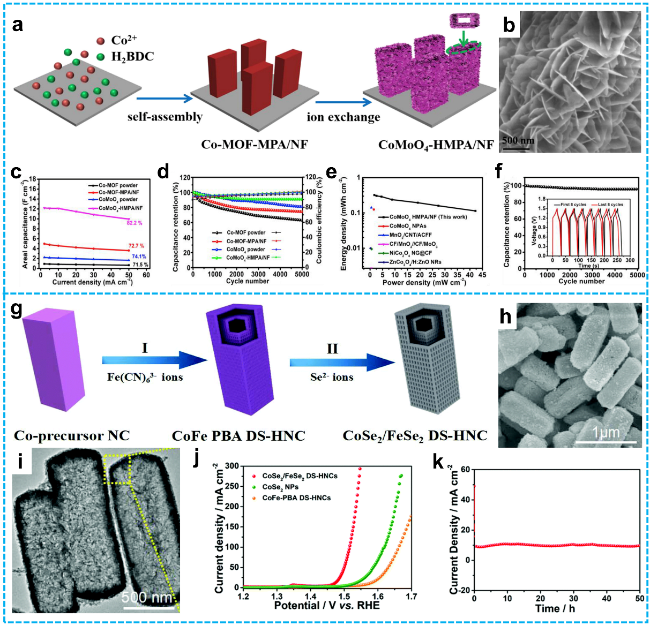
Figure 6 a) Schematic illustration of the synthesis process for CoMoO4-HMPA/NF composite; b) SEM images of CoMoO4-HMPA/NF; c) Areal capacitances versus current densities; d) Cycling performance and coulombic efficiency comparisons among CoMoO4-HMPA/NF, CoMoO4 powder, Co-MOF-MPA/NF, and Co-MOF powder; e) Energy and power density plots of the CoMoO4-HMPA/NF//AC asymmetric supercapacitor (ASC), alongside literature data; f) Cycling stability of the CoMoO4-HMPA/NF//AC ASC device [88]. Copyright 2019, Elsevier. g) Schematic diagram depicting the stepwise ion exchange strategy for synthesizing CoSe2/FeSe2 DS-HNCs; h) SEM, i) TEM images of the as-obtained CoSe2/FeSe2 DS-HNCs; j) LSV curves for OER testing of CoSe2/FeSe2 DS-HNCs, CoSe2 NPs and CoFe-PBA DS-HNCs; k) Chronoamperometric response of CoSe2/FeSe2 DS-HNCs at a constant potential of 1.47 V [56]. Copyright 2019, Royal Society of Chemistry. |
2.3.5 Thermally Induced Matter Repositioning
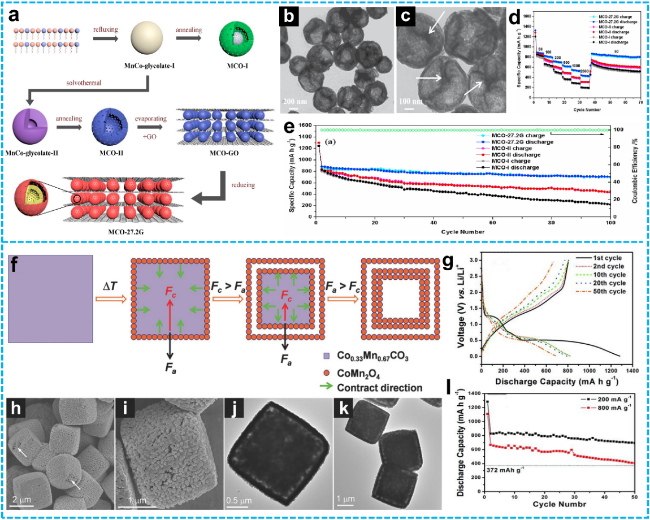
Figure 7 a) Schematic illustration of the typical synthesis routes for yolk-shell MCO-I spheres, mesoporous hollow MCO-II spheres, and MCO-27.2G nanocomposite; b-c) TEM images of mesoporous hollow MCO-II spheres; d) Rate capability of yolk-shell MCO-I spheres, mesoporous hollow MCO-II spheres, and MCO-27.2G nanocomposite electrodes at current densities ranging from 50 to 2000 $mA \cdot g^{-1}$; e) Cycling performance at 200 $mA \cdot g^{-1}$[93]. Copyright 2018, Elsevier. f) Schematic diagram of the synthesis of double-shelled CoMn2O4 hollow microcubes; g) discharge/charge profiles; FESEM (h-i), TEM (j-k) images of double-shelled CoMn2O4 hollow microcubes; l) Cycling stability of double-shelled CoMn2O4 hollow microcubes in the voltage window of 0.01-3.0 V vs. Li/Li⁺ at current densities of 200 and 800 $mA \cdot g^{-1}$ [94]. Copyright 2011, John Wiley and Sons. |
3 Energy Storage and Conversion Applications
Table 2 Comparison of hollow structure preparation methods. |
| Method type | Method name | Advantage | Disadvantage | Typical application areas |
|---|---|---|---|---|
| Hard template method | Hard template | Controllable structure, uniform morphology, wide applicability, and good repeatability | Complicated steps, complex template preparation and removal, possible introduction of impurities, high cost | Catalysis, drug delivery, energy storage |
| Soft template method | Soft template | Adjustable pore structure, suitable for complex multi-level porous materials, mild conditions | Low template stability, sensitivity to reaction conditions, and difficulty in use in multi-component systems | Photocatalysis, battery electrodes, adsorbent materials |
| Template-free method | Selective etching | No need for templates, simple steps, and the ability to prepare multi shell structures | Dependent on the chemical differences of precursors and difficult to control | Catalysis, sensors |
| Ostwald ripening | Template required, controllable number of shell layers, suitable for various metal oxides | Long reaction time and complex kinetic control | Gas sensing, biocatalysis, batteries | |
| Kirkendall effect | Suitable for various materials (metals, oxides, sulfides, etc.), controllable cavity formation | High requirements for diffusion rate and possible formation of non ideal structures | Gas sensing, electrochemical energy storage | |
| Ion exchange | Capable of preparing multi-component compounds with diverse structures, suitable for complex hollow structures | The ion exchange process is difficult to control and may lead to structural collapse | Supercapacitors, electrocatalysts | |
| Thermally induced matter repositioning | Capable of preparing multi shell hollow structures, template free, suitable for multi metal oxides | High temperature treatment may lead to sintering or structural damage | Lithium-ion batteries, supercapacitors |
3.1 Energy Storage
3.1.1 Lithium-ion Batteries
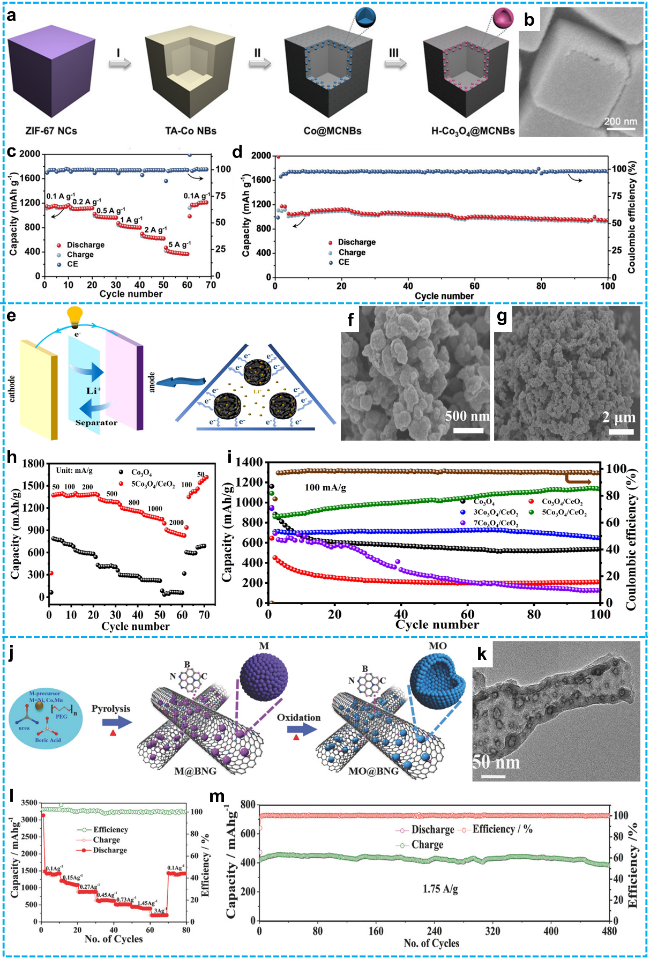
Figure 8 a) Schematic illustration of the formation process of H-Co3O4@MCNBs; b) FESEM images of H-Co3O4@MCNBs; c) Rate capability and coulombic efficiency profiles at different current densities; d) Cycling stability at 0.2 A·g-1 and the associated Coulombic efficiency [110]. Copyright 2020, John Wiley and Sons. e) Schematic of a lithium-ion battery; Li+ shuttles between cathode and anode via separator; Left: overall battery structure with electron flow powering a device; Right: enlarged view of anode showing Li+ and e- movement, key for anode electrochemical reactions during charge-discharge; f-g) SEM images of 5 Co3O4/CeO2; h) rate performance of Co3O4 and 5 Co3O4/CeO2; i) Cycling performance of as-prepared samples at 100 mA·g-1 [111]. Copyright 2021, Elsevier. j) Synthesis schematic of MO@BNG (MO: CoO, Ni2O3, Mn3O4) nanotubes; k) TEM image of CoO@BNG nanotubes of hollow CoO nanoparticle; l) Storage capacity of CoO@BNG nanotubes at various current densities; m) Long-term cycling stability of CoO@BNG at a high current density of 1.75 A·g-1 [109]. Copyright 2018, John Wiley and Sons. |
3.1.2 Lithium-Sulfur Batteries
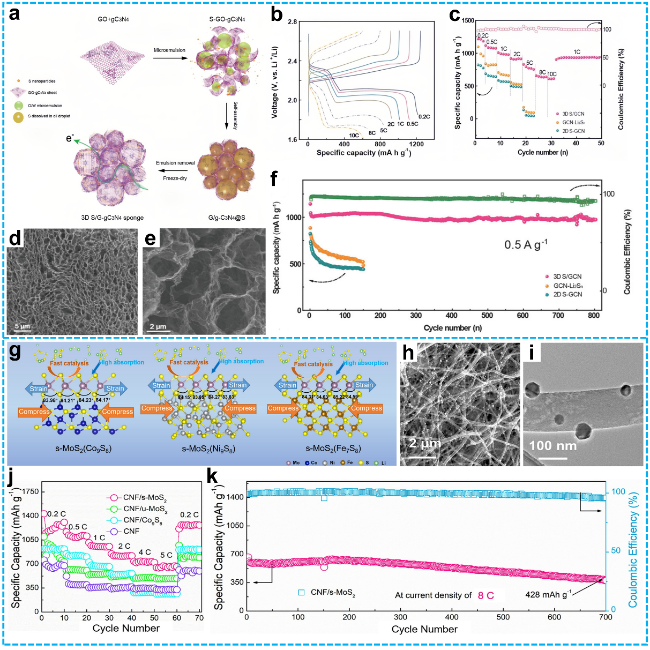
Figure 9 a) Schematic diagram of the preparation process for S/GCN hybrid sponges; b) Galvanostatic charge-discharge (GCD) voltage profiles of the S/GCN cathode at various charge-discharge rates; c) Rate performance of S/GCN, GCN-Li2Sn, and S-GCN electrodes (Coulombic efficiency data correspond to the S/GCN cathode); d) Cross-sectional morphology and e) high-resolution SEM image of S/GCN; f) Long-term cycling stability of S/GCN, GCN-Li2Sn, and S-GCN cathodes at a low current density of 0.5 A·g-1 (0.3 C) [125]. Copyright 2018, John Wiley and Sons. g) Ball-and-stick models depicting strained MoS2 adsorbed on Co9S8, Ni9S8, and Fe7S8 surfaces; h) SEM and i) low-magnification TEM images of CNF/s-MoS2(Co9S8) composites; j) Rate performance of the cells from 0.2 C to 5 C; k) Long-term cycling stability of the CNF/s-MoS2 electrode over 700 cycles at 8 C [130]. Copyright 2022, Elsevier. |
3.1.3 Supercapacitors
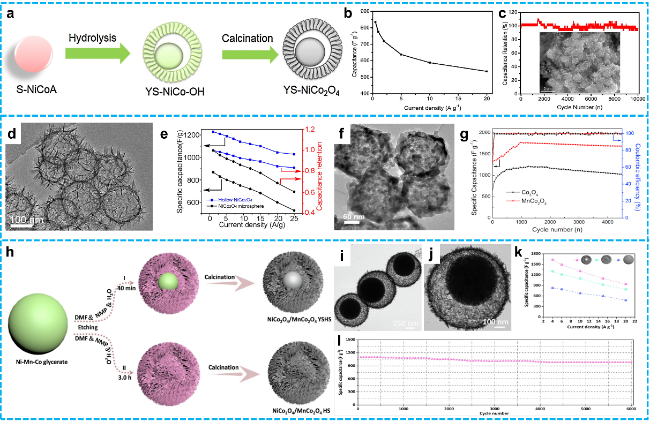
Figure 10 a) Diagram of the formation processes of YS-NiCo2O4; b) the capacitance and c) the cycle life at 10 A·g-1 and SEM image of YS-NiCo2O4 after 10, 000 cycles in inset [144]. Copyright 2018, Elsevier. d) TEM image of hollow NiCo2O4 nanospheres; e) The specific capacitances and capacitance retentions of the hollow NiCo2O4 nanospheres and NiCo2O4 microspheres [145]. Copyright 2018, Elsevier. f) TEM image of the as-obtained nanocage MnCo2O4; g) Coulombic efficiency of the two electrodes at the density of 1 A·g-1 [146]. Copyright 2017, Elsevier. h) Schematic illustration of the process for controllable synthesizing NiCo2O4/MnCo2O4 hollow structures; i-j) TEM images of NiCo2O4/MnCo2O4 YSHS;k) Specific capacitance of NiCo2O4/MnCo2O4 SS, HS and YSHS at current density 4.0-20 A·g-1; l) Cycling performance of NiCo2O4/MnCo2O4 YSHS at 15 A·g-1 over 6000 continuous charge-discharge [99]. Copyright 2023, Elsevier. |
3.2 Energy Conversion
3.2.1 Electrocatalysis
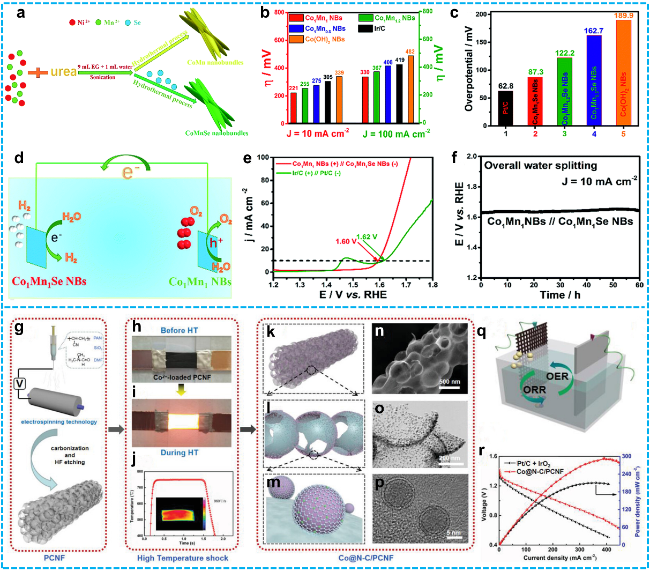
Fig 11. a) Schematic representation of the synthetic methodology for CoMn and CoMnSe nanobundles; b) Overpotential values at current densities of 10 and 100 mA·cm-2 and c) Overpotentials at a current density of 10 mA·cm-2 for Co1Mn1Se NBs, Co1Mn1.2Se NBs, Co1Mn0.8Se NBs, Pt/C, and Co(OH)2 NBs; d) Schematic diagram of the overall water electrolysis process; e) Polarization curves for the Co1Mn1 NBs (+)//Co1Mn1Se NB (−) electrode pair and the Ir/C (+)//Pt/C (−) pair in 1.0 M KOH solution at a scan rate of 5 mV·s-1; f) Continuous chronopotentiometry (CP) measurement of the Co1Mn1 NBs (+)//Co1Mn1Se NB (−) couple over 36 hours [167]. Copyright 2018, Royal Society of Chemistry. g) Schematic representation of the high-temperature (HT) shock synthesis of PCNF; h-i) Photographic sequences depicting the heating procedures employed in the fabrication of Co@N-C/PCNF; j) Temperature profile during the HT shock process, with an inset showing the temperature distribution map; k-m) Structural modeling of distinct components in Co@N-C/PCNF, accompanied by n) SEM, o) TEM, and p) HRTEM images that characterize the selected structural features of the Co@N-C/PCNF composite; q) Schematic illustration of the AZAB configuration; r) Corresponding discharge polarization curves and power density profiles [170]. Copyright 2023, John Wiley and Sons. |
3.2.2 Photocatalysis
Scheme 4 Steps of the photocatalytic reaction process. (I) Light absorption generates electron-hole pairs; (II) Separation of excited charges and transfer of electrons and holes to the photocatalyst surface; (III) Utilization of surface charges for redox reactions. |
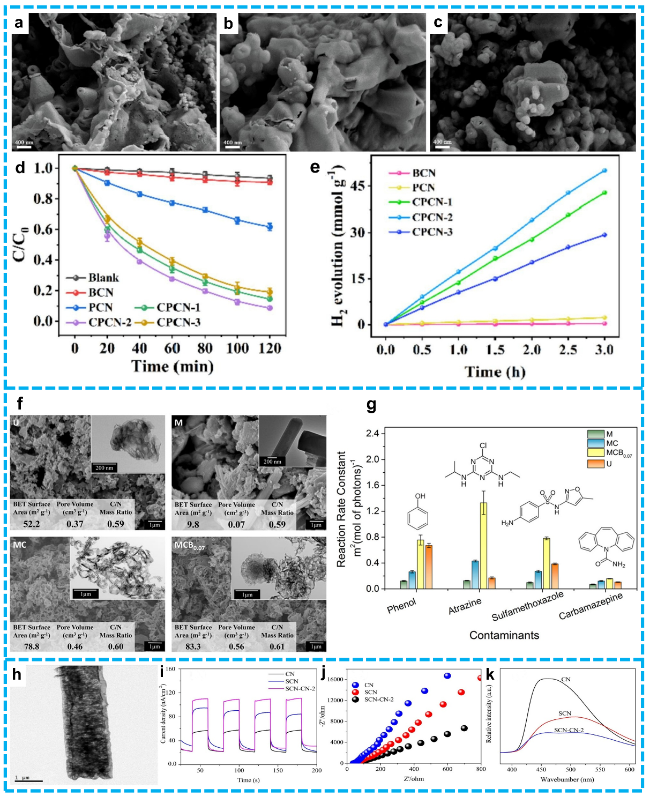
Figure 12 SEM images of a) CPCN-1, b) CPCN-2, c) CPCN-3; d) Photocatalytic degradation of DCF by the as-prepared samples; e) H2 generation of the BCN, PCN and CPCN catalysts [193]. Copyright 2022, Elsevier. f) SEM and TEM (insets) images of g-C3N4 samples U, M, MC, and MCB0.07; g) Photocatalytic degradation rate constants of U, M, MC, and MCB0.07 [194]. Copyright 2016, American Chemical Society. h) TEM images of SCN-CN-2, The photocurrent (i), EIS (j), and PL k) of as-prepared samples [195]. Copyright 2021, Elsevier. |
4. In Situ Testing Techniques Combined with First Principles Calculation in Hollow-structured Materials
4.1 In Situ Irradiation X-ray Photoelectron Spectroscopy (ISI-XPS) Facilitates Exploration of Intrinsic Mechanisms
4.1.1 The ISI-XPS Instrument Fundamental Principles
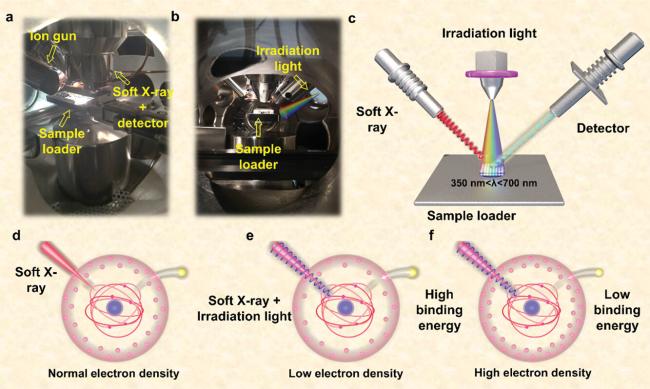
Figure 13 a) Front windows photographs of ISI-XPS device; b) Side windows photographs of ISI-XPS device; c) Schematic illustration of ISI-XPS device; d-f) Effect of electron density on binding energy [207]. Copyright 2022, John Wiley and Sons. |
4.1.2 The Application of ISI-XPS in Hollow-structured Materials
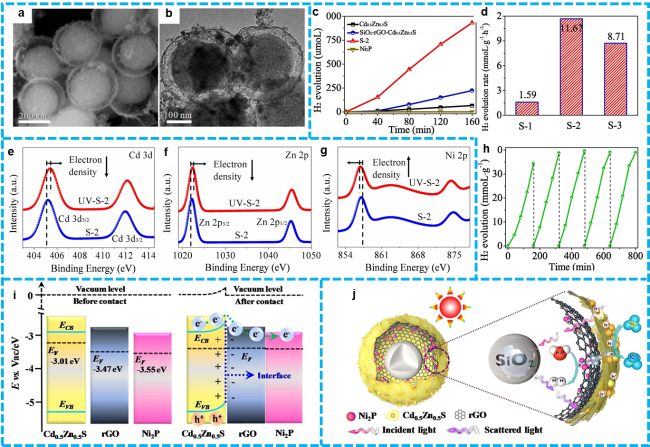
Figure 14 a) The SEM and b) TEM images of as-prepared yolk-shell structural SiO2/Ni2P/rGO/Cd0.5Zn0.5S sample; c) The time dependent H2 evolution over different samples; d) the H2 production rate of different ration composites; h) recycling test over the S-2 composite under visible light irradiation, e-g) High-resolution XPS spectra of the S-2 composite in the dark and under LED irradiation (λ=365 nm): Cd 3d, Zn 2p and Ni 2p; i) Band structure diagram of the Cd0.5Zn0.5S, rGO and Ni2P; j) Photocatalytic mechanism of H2 generation in the yolk-shell structural SiO2/Ni2P/rGO/Cd0.5Zn0.5S nanoreactor [208]. Copyright 2021, Elsevier. |
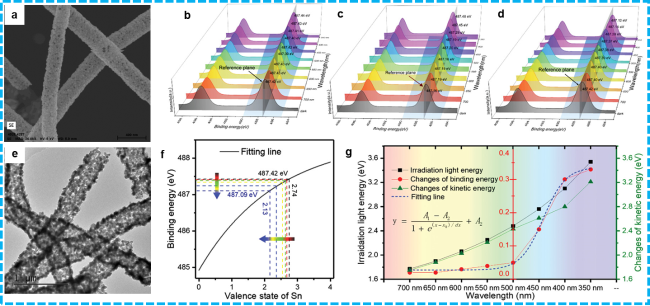
Figure 15 a) The SEM image of g-C3N4/SnO2; e) HRTEM of g-C3N4/SnO2; b) ISI-XPS result of g-C3N4/SnO2 before charge compensation; c) ISI-XPS result of SnO2; d) ISI-XPS result of g-C3N4/SnO2 after charge compensation; f) The fitting curve of the relation between binding energy and valence state of Sn element in g-C3N4/SnO2; g) Relationship of irradiation light energy, changes of binding energy, and kinetic energy of Sn 3d5/2 in g-C3N4/SnO2. [209]. Copyright 2023, Elsevier. |
4.2 In Situ Surface Attenuated Total Reflection Fourier Transform Infrared Spectroscopy (ATR-FTIR) Combining with First Principles Calculation Facilitates Exploration of Intrinsic Mechanisms.
4.2.1 ATR-FTIR Fundamental Principles
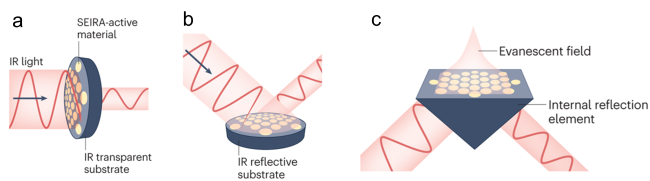
Figure 16 Schematic illustration of the three primary SEIRAS techniques: a) transmission mode, b) external reflection mode, and c) attenuated total reflection (ATR) mode [221]. |
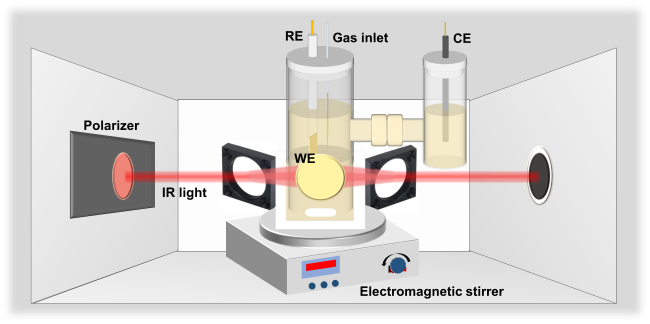
Figure 17 Schematic diagram of an in situ electrochemical ATR-FTIR setup equipped with stirring capability [222]. |
4.2.2 The Application of SEIRAS Combining with First Principles Calculation for Elucidation of Catalytic Mechanisms
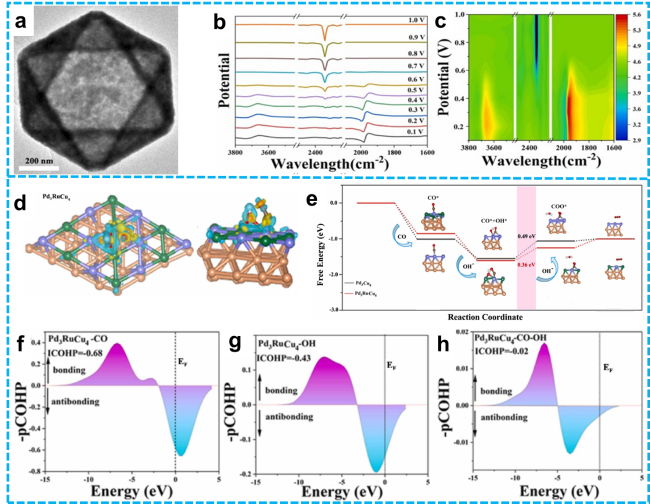
Figure 18 a) TEM images of Pd3RuCu4 NCs; In situ FTIR spectral analysis of adsorbed species on Pd3RuCu4 NCs: b) FTIR spectra and c) contour maps of OH, CO, and CO2 adsorption; d) Local charge density difference of CO co-adsorbed with OH; e) Energy profile comparison for CO oxidation to CO2; f) Crystal orbital Hamilton population (pCOHP) analysis: g) CO adsorption, h) OH adsorption. [227]. Copyright 2025, Elsevier. |
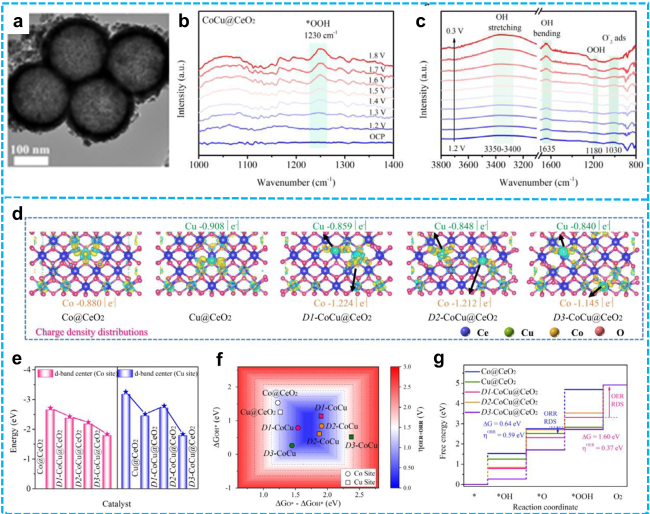
Figure 19 a) TEM images of CoCu@CeO2; In situ FT-IR spectra of CoCu@CeO2 for b) OER and c) ORR; Theoretical calculation: d) Charge density distributions and Bader charge at the Co and Cu sites; e) the d-band center at the Co and Cu sites for Co@CeO2, Cu@CeO2, D1-CoCu@CeO2, D2-CoCu@CeO2, and D3-CoCu@CeO2; f) Calculated total theoretical overpotential (𝜂total) volcano plot with ΔGO*−ΔGOH* and ΔGOH* as descriptors; g) Theoretical overpotential of the catalyst for OER (at Co site) and ORR (at Cu site) for Co@CeO2, Cu@CeO2, D1-CoCu@CeO2, D2-CoCu@CeO2, and D3-CoCu@CeO2 [230]. Copyright 2025, John Wiley and Sons. |
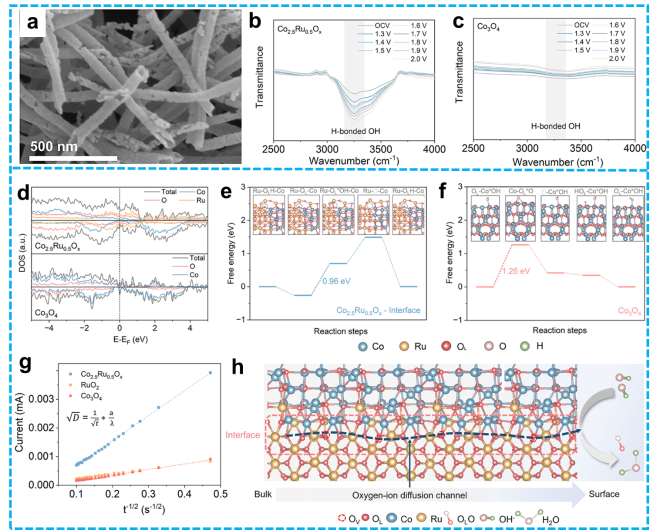
Figure 20 a) SEM image of hollow-nanotube hybrid Co2.5Ru0.5Ox composites; b-c) In situ FTIR spectra of hollow-nanotube hybrid composites; d) DOS diagrams of hybrid Co2.5Ru0.5Ox and Co3O4; Free energy diagram of the LOM pathway for e) hybrid Co2.5Ru0.5Ox with Co-O-Ru interfaces and f) Co3O4; g) Chronoamperometry test of synthesized samples to calculate oxygen ion diffusion coefficients; h) Mechanistic diagram of lattice oxygen-ion diffusion at the Co3O4-x-RuO2-x interface [233]. Copyright 2025, ACS Publications. |
5. Summary and Outlook
Figure 21 Outlook for future research on hollow structures. |