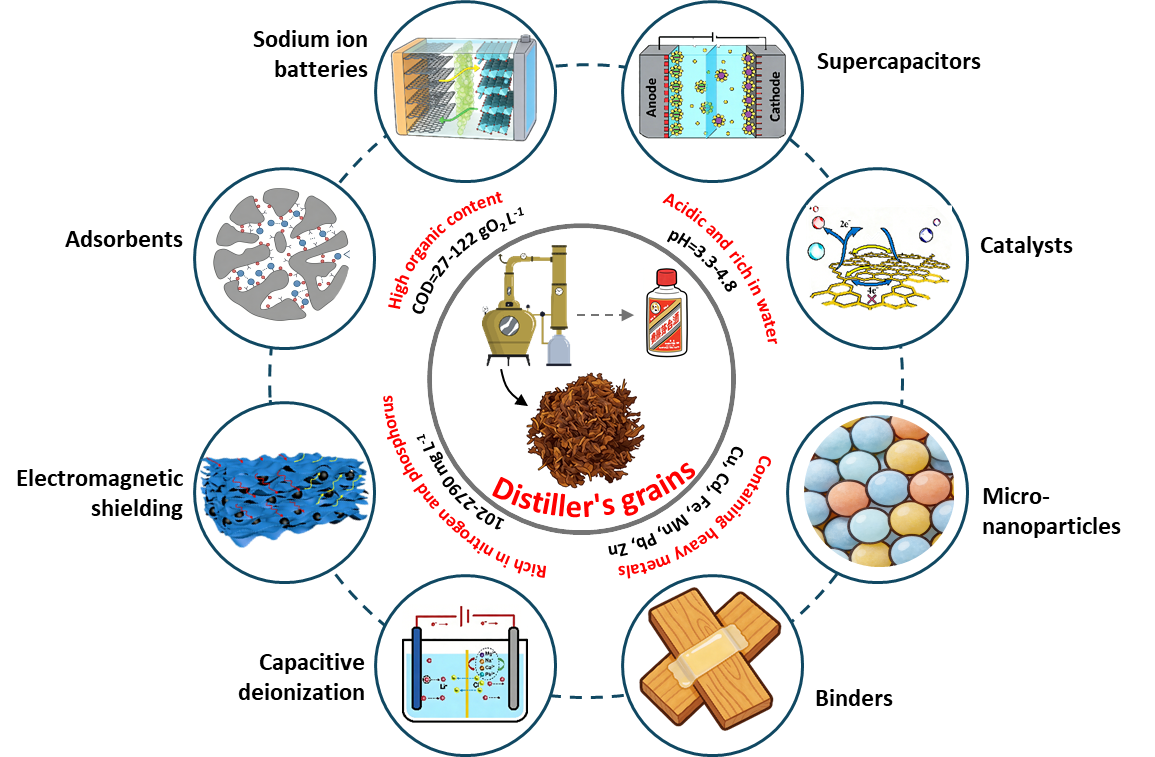
1. Introduction
1.1 Origin of distiller's grains
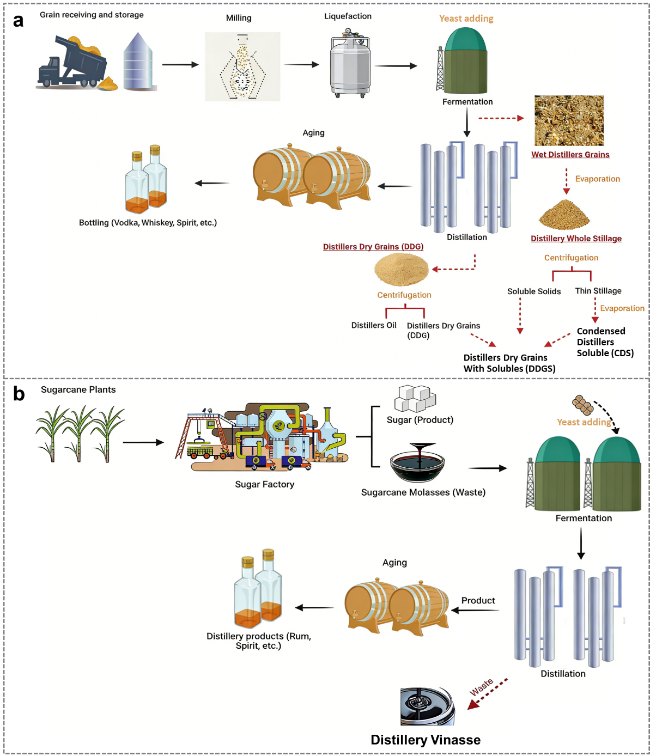
Figure 1. (a) Distillery industries and by-products (Distiller's Grains and Distillery Stillage). (b) Distillery industries and by-products (Distillery Vinasse) [1]. |
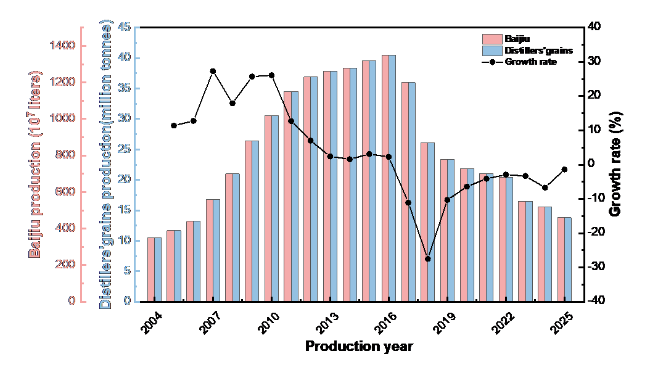
Figure 2. Annual output of Chinese Baijiu and distiller's grains, between 2004 and 2025(Data source: National Bureau of Statistics of China). |
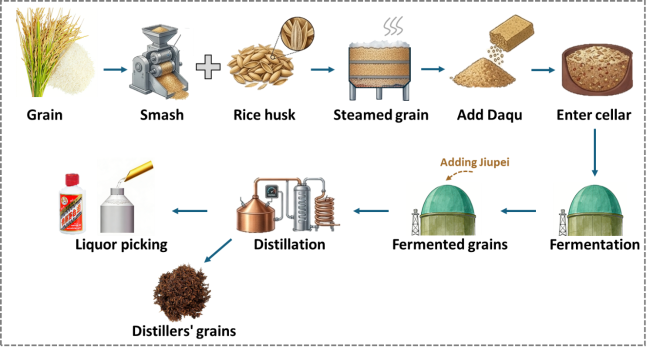
Figure 3. The production process of Chinese Baijiu. |
1.2 Basic properties of distiller's grains
| Parameter | Unit | Sugarcane Juice Vinasse | Sugarcane Molasses Vinasse | Grape Vinasse | Beet Vinasse | Sweet Sorghum Vinasse | Agave Vinasse |
|---|---|---|---|---|---|---|---|
| pH | - | 3.3-4.9 | 4.5-4.8 | 2.9-4.2 | 4.3-5.3 | 4.5 | 3.4-3.8 |
| BOD | g O2 L-1 | 5-17 | 39 | 14-19 | 27-45 | 46 | 21-33 |
| COD | g O2 L-1 | 27-30 | 85-95 | 26-50 | 55-91 | 80 | 56-122 |
| Total Solids | kg L-1 | 27-81 | - | - | - | - | 27-94 |
| Total Nitrogen | mg L-1 | 102-628 | 153-1230 | 105-650 | - | 800 | - |
| Total Phosphorus | mg L-1 | 71-130 | 1-190 | 65-118 | 41 | 1990 | - |
| Potassium | kg L-1 | 1.7-2 | 4.9-11 | 0.12-0.8 | 10 | - | 0.24-0.34 |
| Sulfate | mg L-1 | 1356 | 1500-3480 | 120 | 3500-3720 | - | 308-947 |
| Phenols | mg L-1 | 450-469 | 34 | 29-474 | 450 | - | 478-542 |
| Cu | mg L-1 | 0.35-0.67 | 0.3-1.7 | 0.2-3.3 | - | 37 | 0.4-4 |
| Cd | mg L-1 | - | 0.04-1.36 | 0.05-0.08 | <0.011 | - | 0.01-0.2 |
| Fe | mg L-1 | 16 | 12.8-157.5 | 0.001-0.077 | - | 317 | 35.2-45 |
| Mn | mg L-1 | 2.7-7.8 | 0.9-5 | - | 3.9 | - | - |
| Pb | mg L-1 | 0.04-0.44 | 1-8.8 | 0.55-1.34 | <0.011 | - | 0.065-0.5 |
| Zn | mg L-1 | 0.43-7.5 | 3.1-11.8 | - | 13 | - | - |
1.3 Traditional applications of distiller's grains
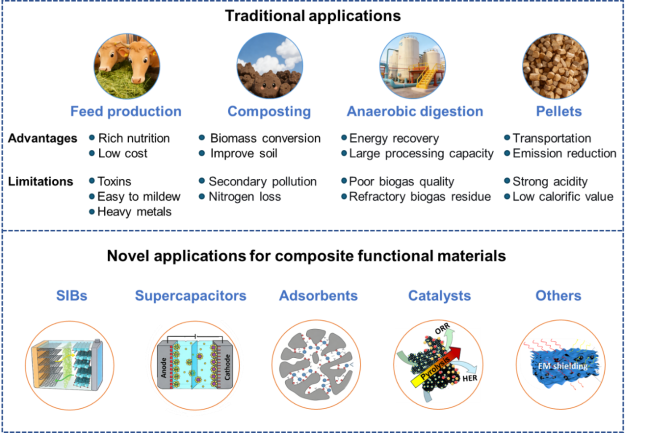
Figure 4. Utilization of distiller's grains. |
1.3.1 Feed production
1.3.2 Composting
1.3.3 Anaerobic digestion
1.3.4 Pellets
2. Advanced applications of distiller's grains
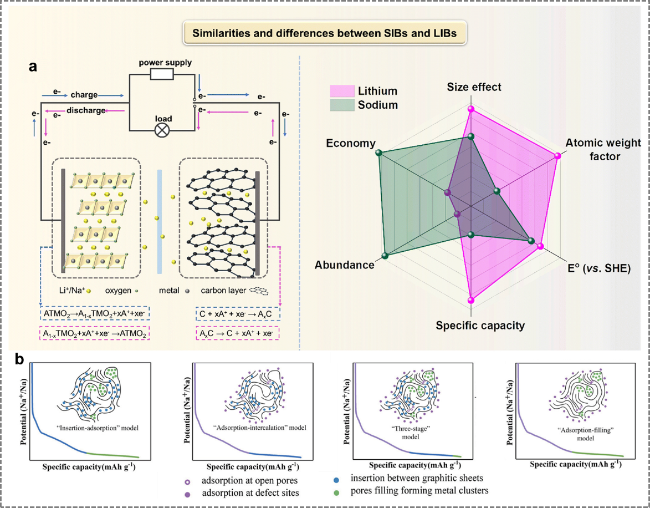
2.1 Sodium-ion batteries
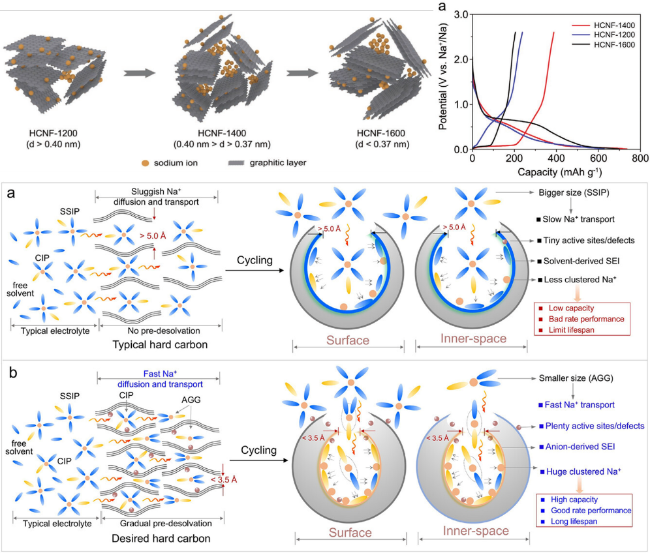
Figure 6. The influence of the aperture of the closed pore on the sodium-ion storage performance of HCs [67]. |
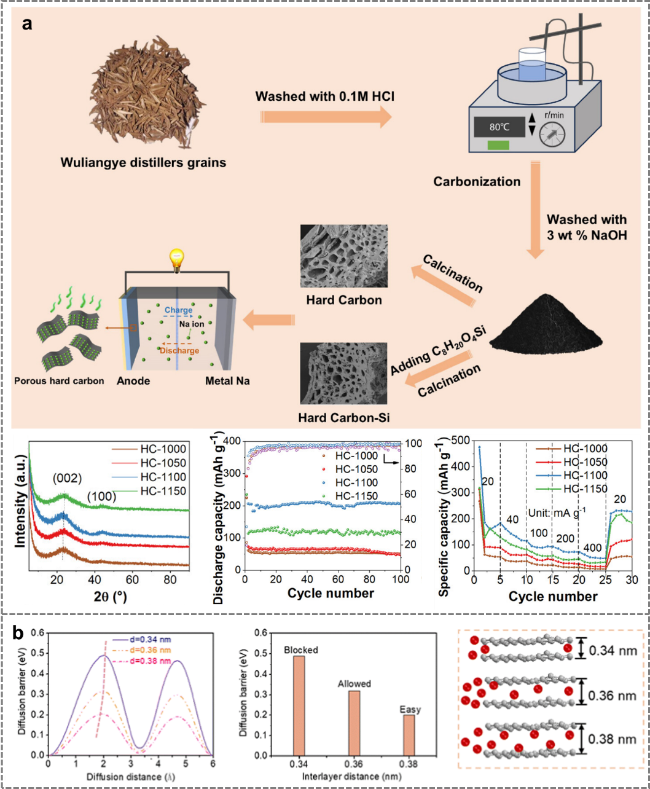
Figure 7. (a) Preparation of distiller's grains-derived hard carbon by two-step carbonization combined with silicon doping for application in sodium-ion batteries [69]. (b) Density functional theory (DFT) calculations elucidate the mechanistic role of interlayer spacing in governing sodium-ion diffusion kinetics [69]. |
2.2 Supercapacitors
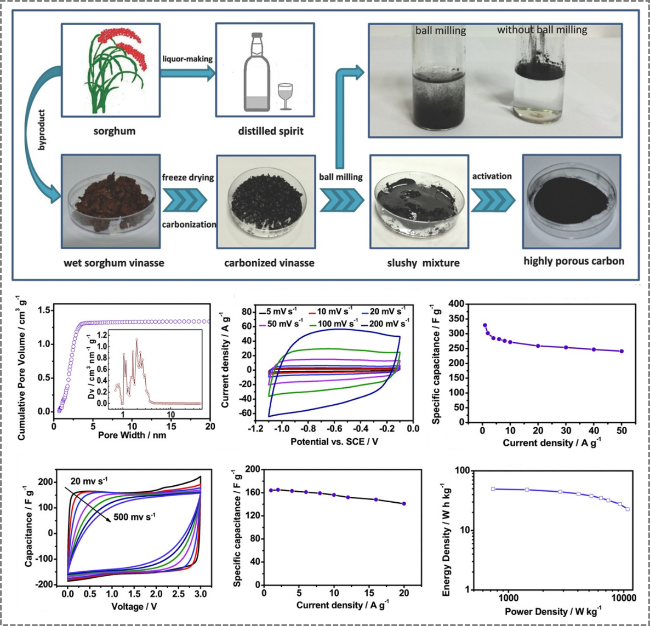
Figure 8. Preparation of distiller's grains-derived porous carbon by two-step KOH activation for application in supercapacitors [87]. |
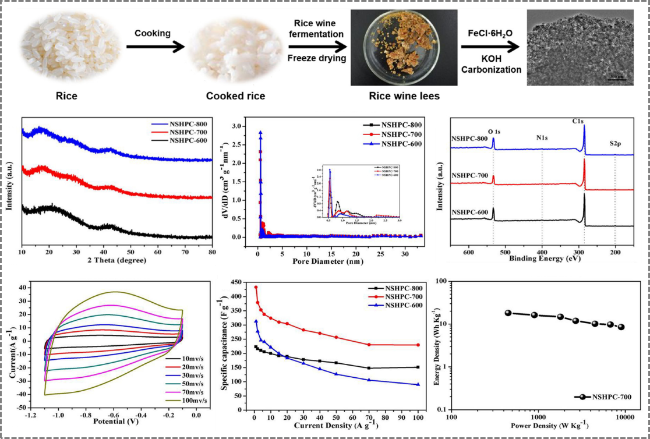
Figure 9. Preparation of distiller's grains-derived porous carbon by synergistic KOH activation and FeCl3·6H2O catalytic activation for application in supercapacitors [93]. |
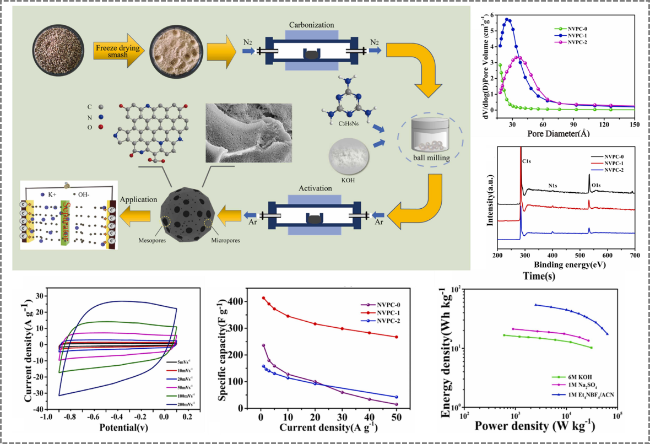
Figure 10. Preparation of nitrogen-doped distiller's grains-derived porous carbon by synergistic KOH activation and melamine doping for application in supercapacitors [99]. |
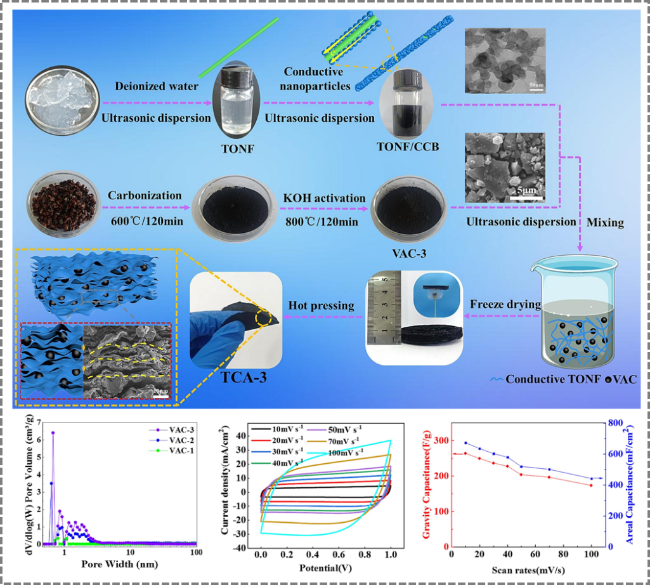
Figure 11. Preparation of conductive TONF/CCB/VAC (TCA) composite material by two-step KOH activation for application in supercapacitors [103]. |
Table 2. Distiller's grains-derived porous carbons prepared by different methods for supercapacitors. |
| Preparation method | Porous carbons | Specific surface | Electrolyte | Current density (A g-1) | Capacitance (F g-1) | Cycling stability | Energy density (Wh kg-1) | Power density (W kg-1) | Ref. |
|---|---|---|---|---|---|---|---|---|---|
| KOH activation | HPC-K750 | 3047 | 2 M KOH | 1/50 | 329/240 | 88% after 10000 cycles | 49.5 | 737 | [81] |
| DG-5-6 | 1027 | 6 M KOH | 1 | 345 | 96.9% after 5000 cycles | 12.2 | 6979.7 | [82] | |
| NaOH activation | SFPC-A13 | 3051 | 6 M KOH | 0.5 | 354 | - | - | - | [95] |
| Hydrothermal carbonization | AC | 479 | 1 M Na2SO4 | 1/5 | 102/70 | 92.3% after 5000 cycles | 9 | 4000 | [86] |
| Transition metal introduction | NSHPC-700 | 1357 | 6 M KOH | 1/10 | 434/305 | 92.3% after 60000 cycles | 17.8 | 450 | [87] |
| Heteroatom doping | NVPC-1 | 3443 | 6 M KOH | 1 | 413 | 107% after 10000 cycles | 10.3 | 30800 | [93] |
| Composite material | TCA | - | 1 M Li2SO4 | 672 mF cm-2 | 263 | 100% after 12000 cycles | 23.8 | 5700 | [103] |
2.3 Adsorbents
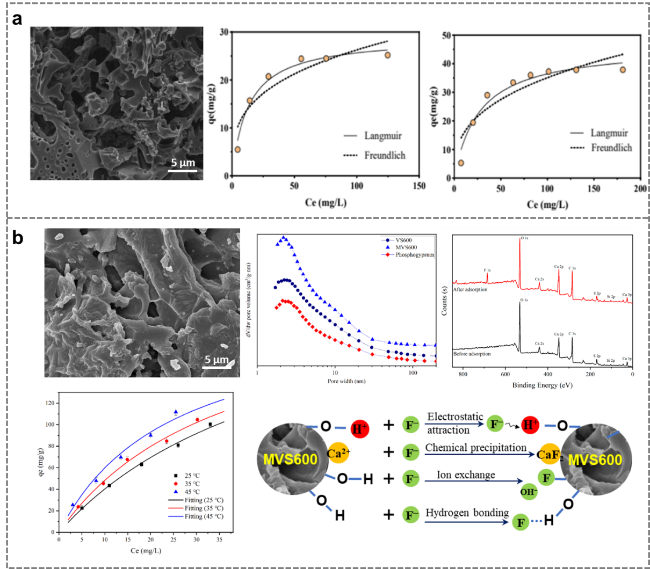
Figure 12. Preparation of distiller's grains-derived activated carbon by direct carbonization for application in pollutant adsorption. (a) Adsorption of tetracycline and ciprofloxacin by vinegar lees-derived activated carbon [109]. (b) Adsorption of fluoride by distiller's grains shell-derived activated carbon [110]. |
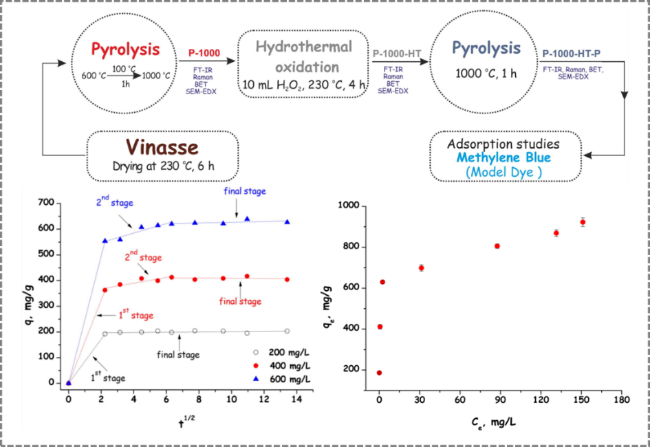
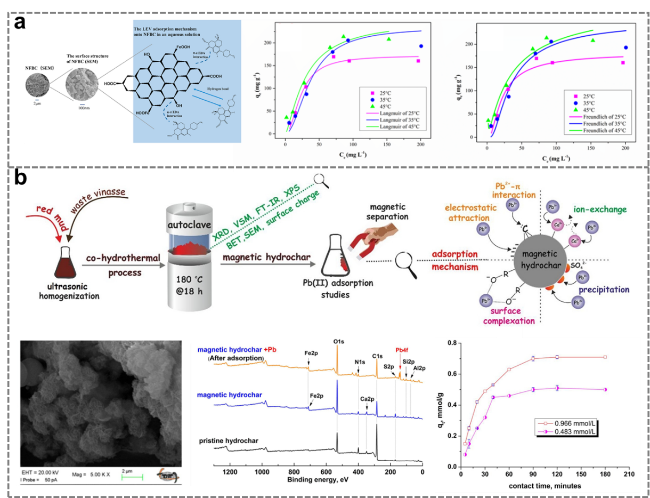
Figure 13. Preparation of distiller's grains-derived activated carbon by hydrothermal treatment for application in methylene blue adsorption [117]. |
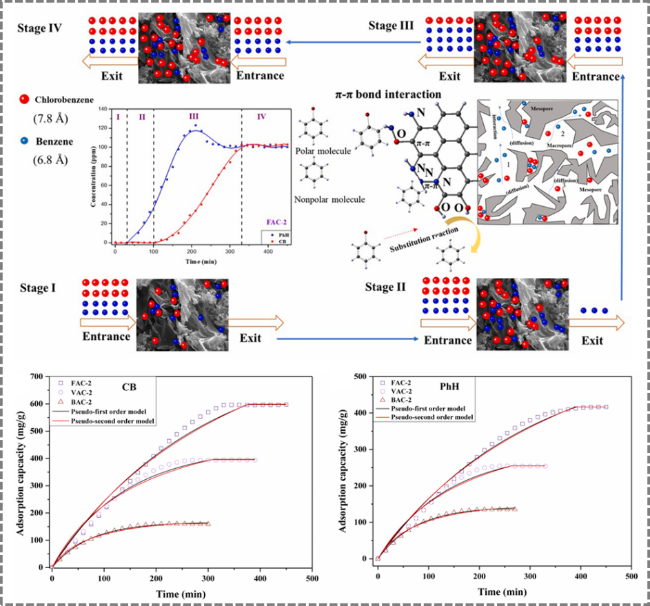
Figure 14. Preparation of distiller's grains-derived activated carbon by chemical activation for application in chlorobenzene (CB) and benzene adsorption [119]. |
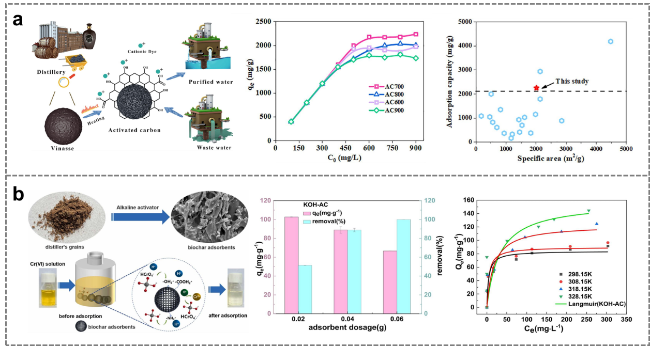
Figure 15. Preparation of distiller's grains-derived activated carbon by pre-carbonization combined with chemical activation for application in pollutant adsorption. (a) Adsorption of methylene blue by distiller's grains-derived activated carbon [122]. (b) Adsorption of Chromium (VI) by distiller's grains-derived activated carbon [123]. |
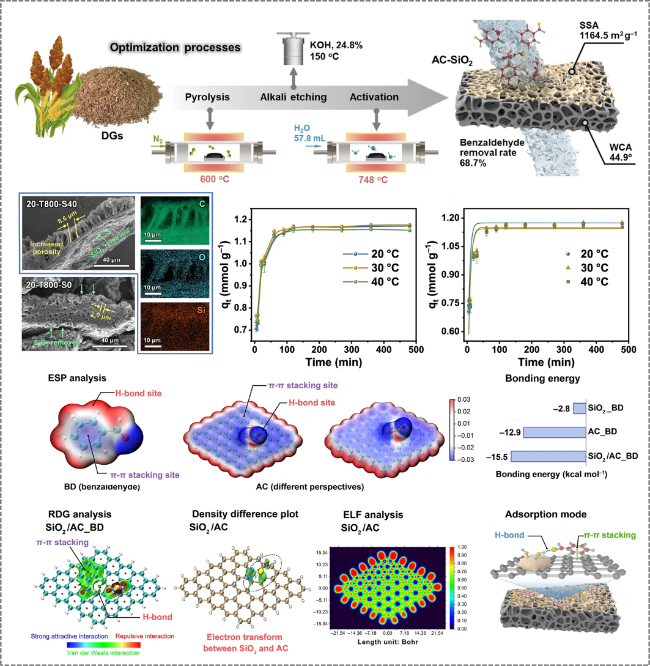
Figure 16. Preparation of distiller's grains-derived activated carbon by physicochemical activation for application in benzaldehyde adsorption [129]. |
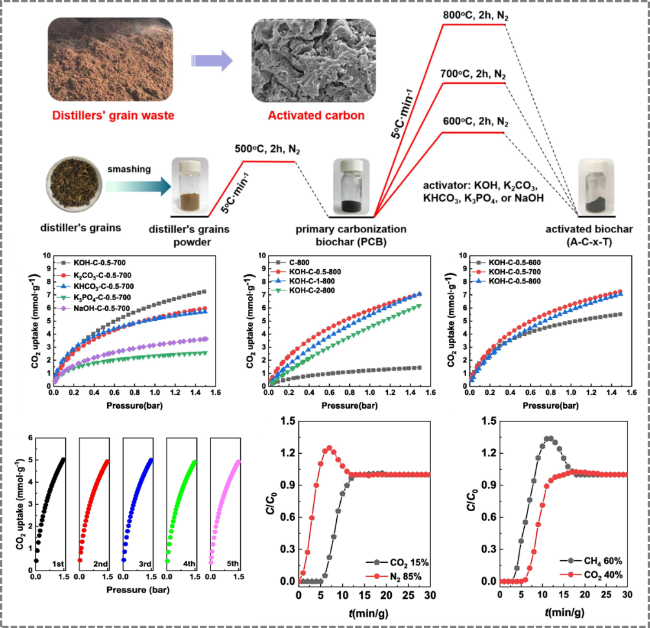
Figure 18. Distiller's grains-derived activated carbon for gas adsorption and separation technology [142]. |
Table 3. Modification methods and adsorption mechanisms of distiller's grains-derived adsorbents. |
| Preparation/modification method | Main active sites introduced | Target pollutant | Main adsorption mechanism | Adsorption capacity (example) |
|---|---|---|---|---|
| Direct carbonization | Porous carbon skeleton | Antibiotic | Physical adsorption, electrostatic attraction | 29-46 mg g-1 |
| Hydrothermal treatment | Rich oxygen-containing functional groups (-OH, -COOH) | Dyestuff | Physical adsorption, electrostatic adsorption, π - π interaction | 909 mg g-1 |
| Chemical activation | High specific surface area and developed microporous structure | Heavy metal | Physical adsorption, electrostatic adsorption | 29.5 mg g-1 |
| Pre-carbonization | Preliminary carbon skeleton, partial pores | Dyes, heavy metals | Physical adsorption, electrostatic adsorption | 144.5 mg g-1 |
| Precombustion | Ash and inorganic mineral components | dyestuff | Physical adsorption, electrostatic adsorption, π - π interaction | 2276.3 mg g-1 |
| Physical activation | Abundant micropores and mesopores | Hydrophobic turbidity | Physical adsorption and hydrophobic interaction | 765.7-1001.0 mg g-1 |
| Physical-chemical combined activation | High specific surface area and rich functional groups | Volatile organic compounds | Physical adsorption, π - π interaction, hydrogen bond | 1.08 mmol g-1 |
| Heteroatom doping | N, O functional group, changing surface charge | Phenol | Physical adsorption, electrostatic adsorption, hydrogen bonding | - |
| Template method | Ordered pore structure (mesoporous) | Drug contaminants | Physical adsorption, electrostatic adsorption | 406-418 mg g-1 |
| Magnetic component modification | Fe3O4/γ - Fe2O3 nanoparticles | Antibiotics, heavy metals | Physical adsorption, electrostatic adsorption, π - π interaction, hydrogen bond | 172 mg g-1 |
2.4 Catalysts
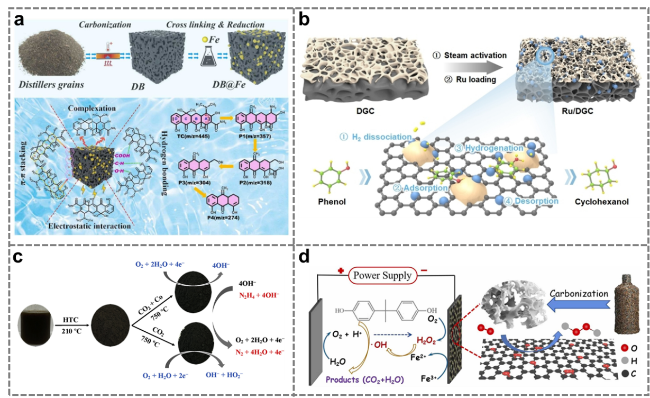
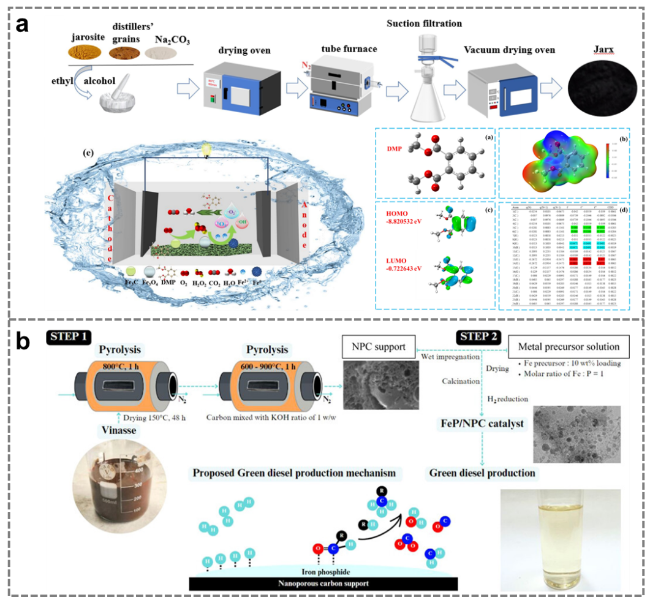
Figure 19. (a) Catalytic removal of tetracycline by distiller's grains-derived catalyst [143]. (b) Catalytic conversion of phenol to cyclohexanol by a distiller's grains-derived catalyst [144]. (c) Catalysis of the oxygen reduction reaction (ORR) by a distiller's grains-derived catalyst [145]. (d) Degradation of organic pollutants by a distiller's grains-derived electro-fenton catalyst [146]. |
Table 4. Economic analysis table of distiller's grains-derived catalysts vs. traditional catalysts [160-163]. |
| Materials | Cost | Energy consumption | Life cycle carbon emissions | Contamination during preparation | Disposal | Comprehensive potential |
|---|---|---|---|---|---|---|
| Baijiu distiller's grains-derived catalyst | CNY 200-300 per ton | Hydrothermal carbonization: 500-800 kWh per ton High-temperature activation: 1000-1500 kWh per ton | 1-2.5 tons CO2e per ton | Waste water and waste gas | Low toxicity | High |
| Traditional catalysts (such as the noble metal Pt/C) | CNY 2-4 million per ton | High-temperature synthesis and processing: 3,000-5,000 kWh per ton | 8-20 tons CO2e per ton | Toxic chemicals | Hazardous waste | Medium |
2.5 Other applications
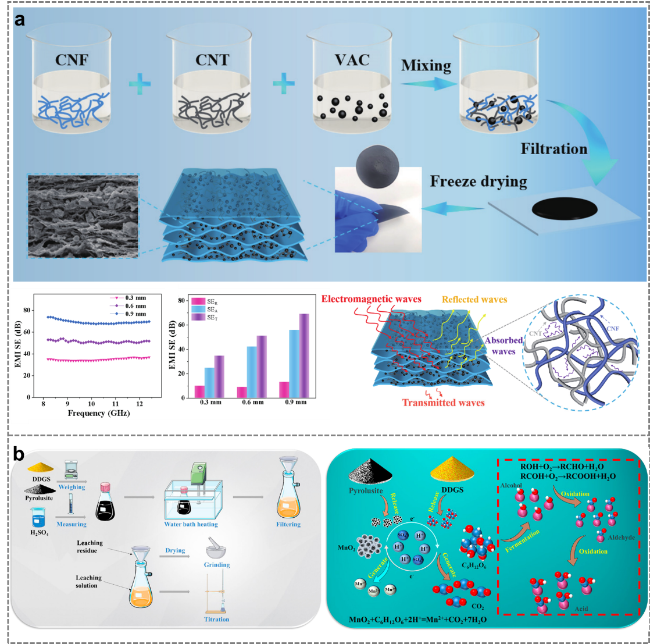
2.5.1 Electromagnetic shielding
2.5.2 Leaching manganese ore
2.5.3 Capacitive deionization
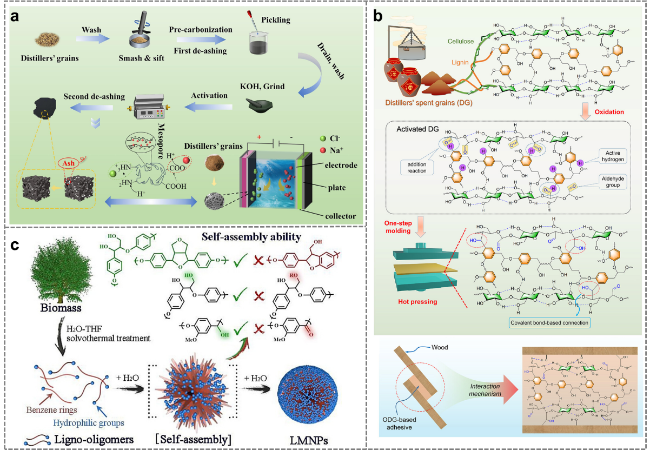
2.5.4 Binders
2.5.5 Micro-nanoparticles
2.5.6 Phase change materials
3. Conclusions
4. Perspectives
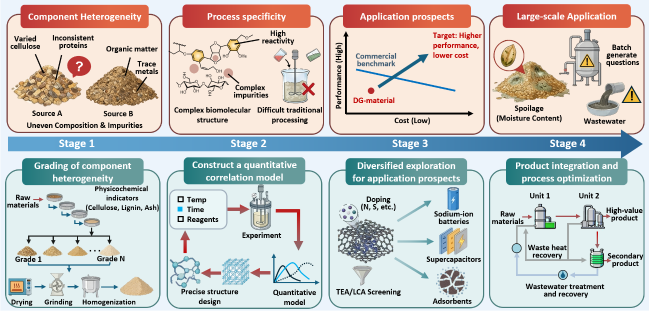
Figure 23. Prospective roadmap of distiller's grains-derived functional materials and composite functional materials. |