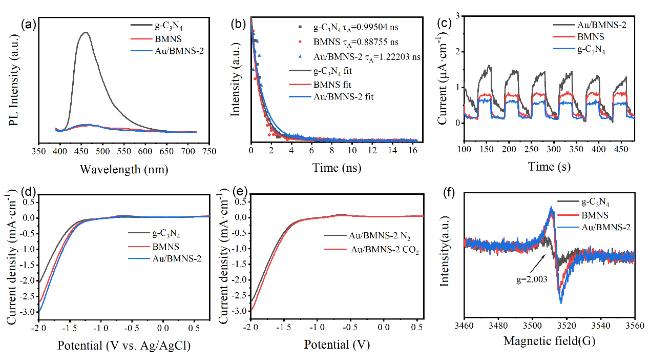
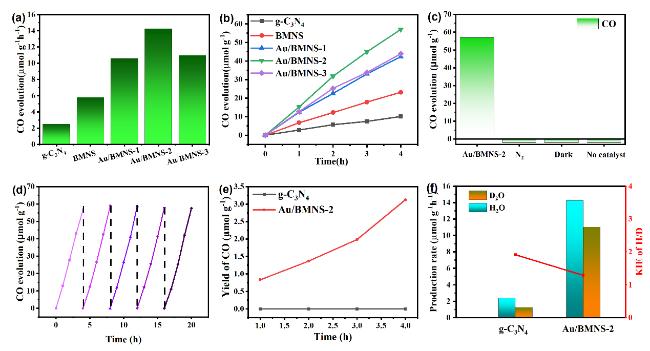
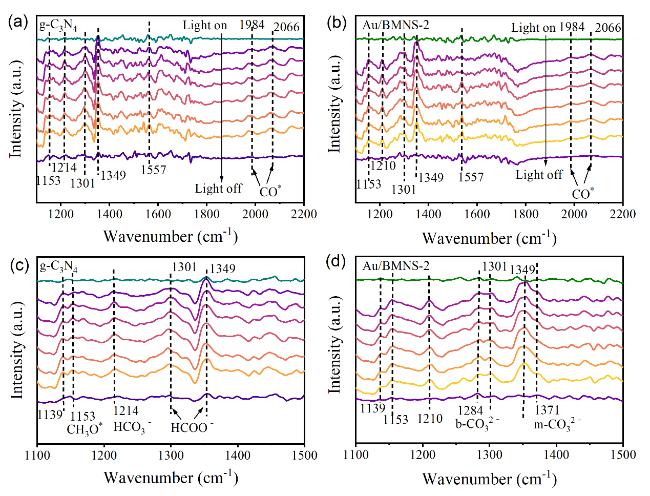
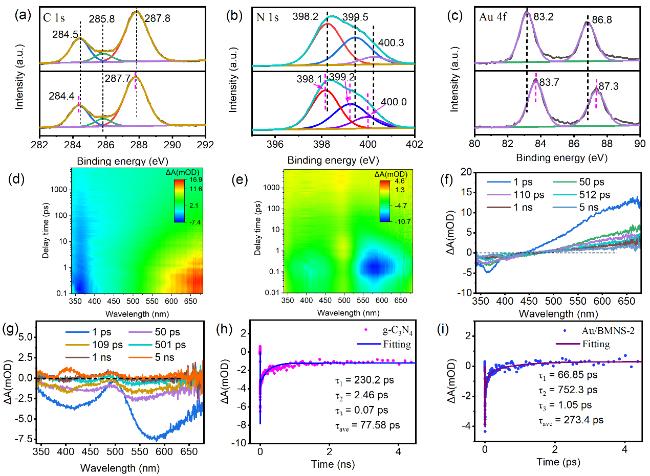
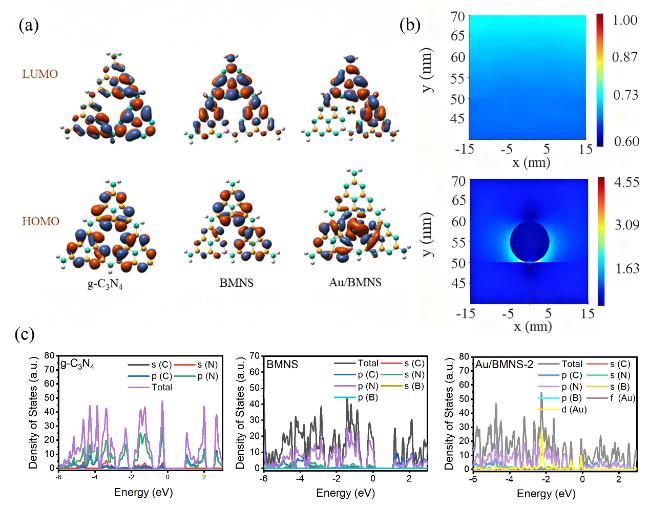
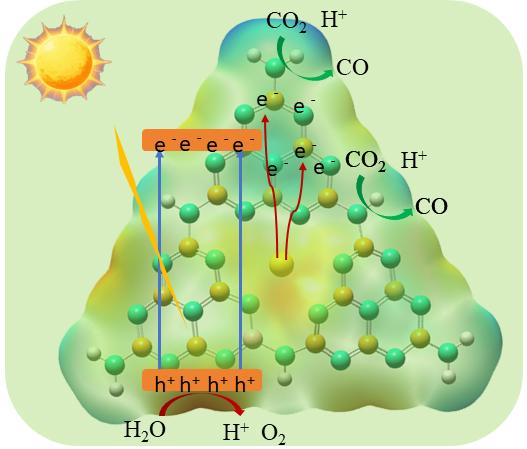
Systematic control experiments were conducted to investigate the photocatalytic stability and the origin of the CO product over the Au/BMNS-2 catalyst. When the reaction was carried out under a N
2 atmosphere with all other conditions unchanged, no CO was detected, effectively excluding the possibility of CO generation from the decomposition of intrinsic carbon-containing species within the catalyst. Additionally, control experiments carried out under otherwise identical conditions, either in the absence of a catalyst (blank test) or in the dark, both resulted in negligible CO production (
Fig. 6c). In addition, a
13C isotope labeling experiment was conducted to unequivocally confirm the carbon source of the observed products (Fig. S3). The detection of
13CO in the final product clearly demonstrates that the generated CO originates from the photoreduction of CO
2, rather than from any carbon-containing contaminants. These results unambiguously demonstrate that CO formation requires the simultaneous presence of three key components: (1) CO
2 as the carbon source, (2) the Au/BMNS-2 catalyst, and (3) light irradiation. To confirm that H
2O rather than any organic component acts as the sacrificial agent, we monitored the evolution of O
2, the oxidation product of H
2O, using a thermal conductivity detector (Fig. S4). A clear O
2 signal was observed, corresponding to a yield of 26.46 μmol·g
-1 over Au/BMNS-2 within 4 hours. The measured molar ratio of CO to O
2 was 2.15, which closely matches the theoretical value of 2. This agreement strongly confirms that CO
2 reduction proceeds via concomitant oxidation of H
2O. Furthermore, the catalyst exhibited excellent recyclability, maintaining a consistent CO evolution rate over five consecutive photocatalytic cycles (
Fig. 6d). No significant deactivation was observed, indicating high structural and photochemical stability under prolonged reaction conditions. Moreover, the Au/BMNS-2 catalyst after five reaction cycles (Au/BMNS-Re) was characterized by XRD and BET analysis (Fig. S5). The XRD pattern of Au/BMNS-Re closely matches that of the fresh Au/BMNS-2, with only a slight reduction in diffraction peak intensity, indicating excellent structural stability and catalytic durability. Furthermore, BET measurements were carried out to assess any changes in specific surface area before and after the reaction. Notably, the specific surface area of Au/BMNS-Re remains nearly identical to that of the pristine Au/BMNS-2 (Fig. S5b and Table S2), further confirming the robustness of the catalyst’s porous structure during the photocatalytic process. These post-reaction characterization results are fully consistent with the observed stability in catalytic activity over multiple cycles. Collectively, these findings confirm that the detected CO originates exclusively from the photocatalytic reduction of CO
2 and highlight the robust stability and reliability of the Au/BMNS-2 catalyst.