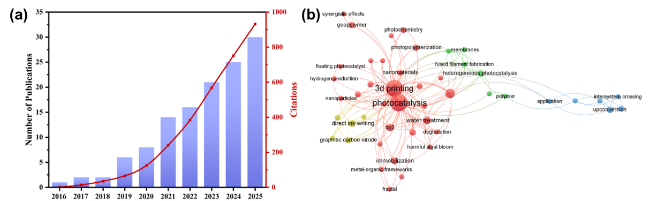
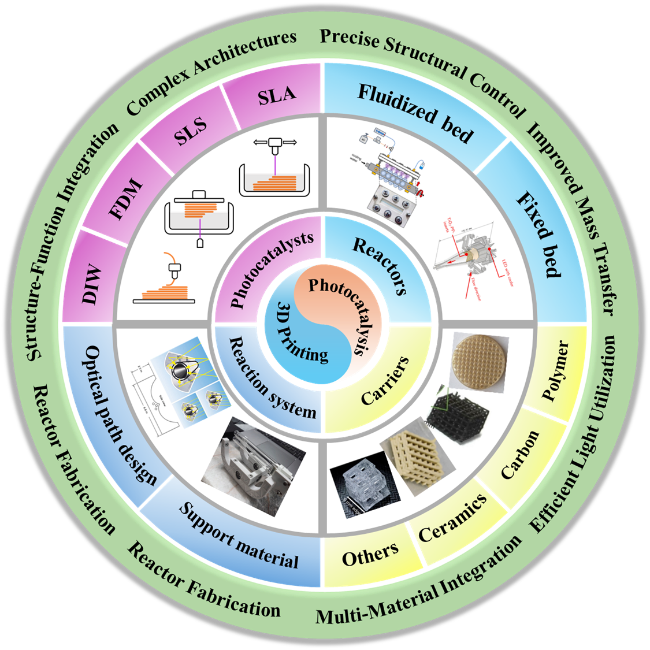
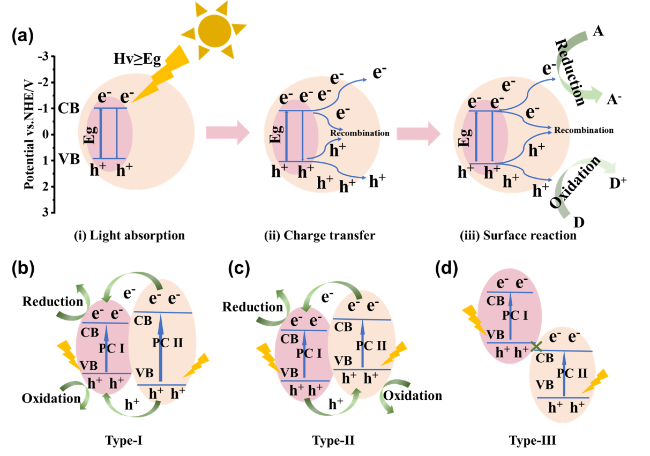
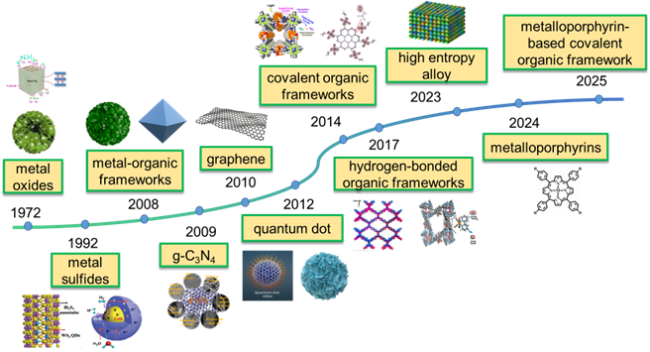
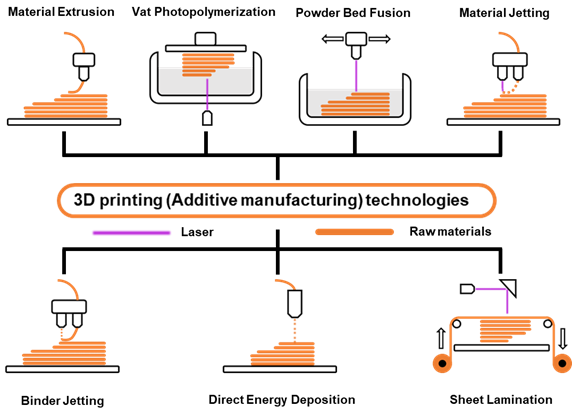
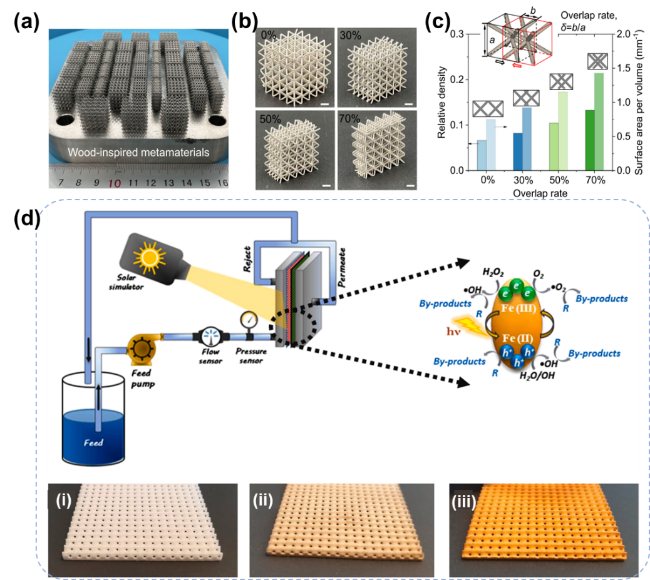
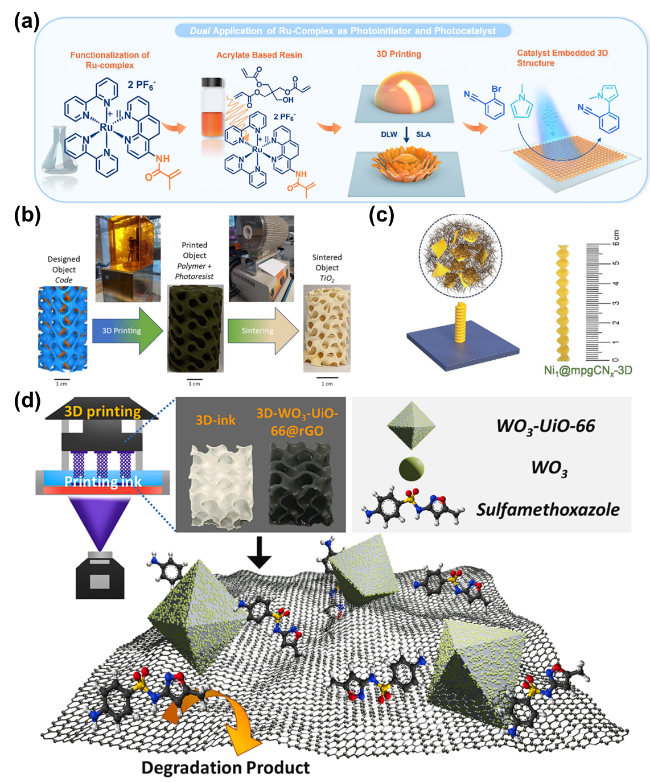
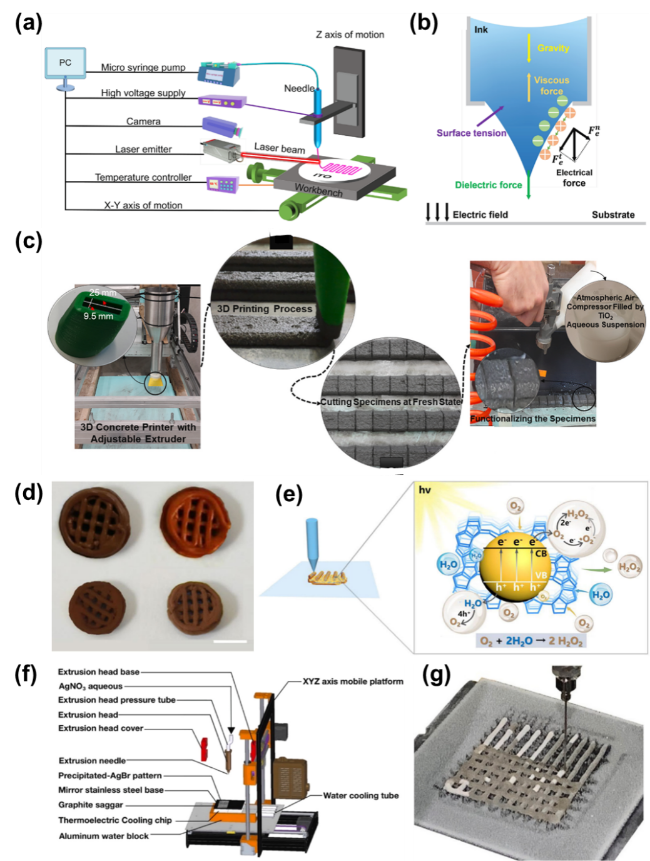
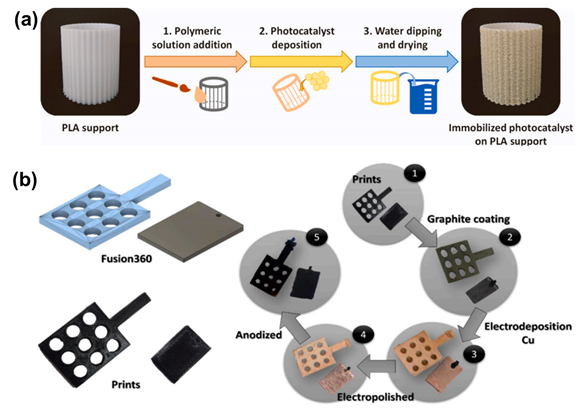
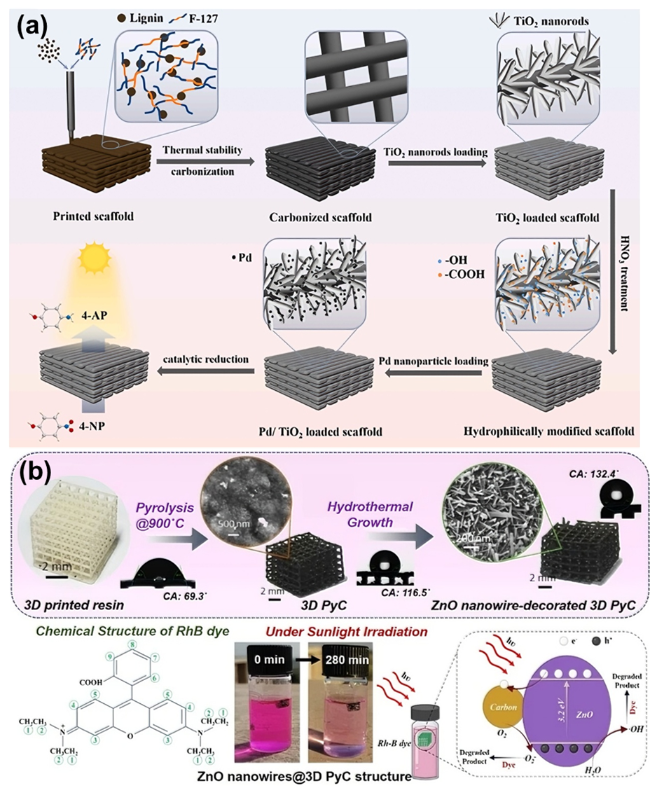
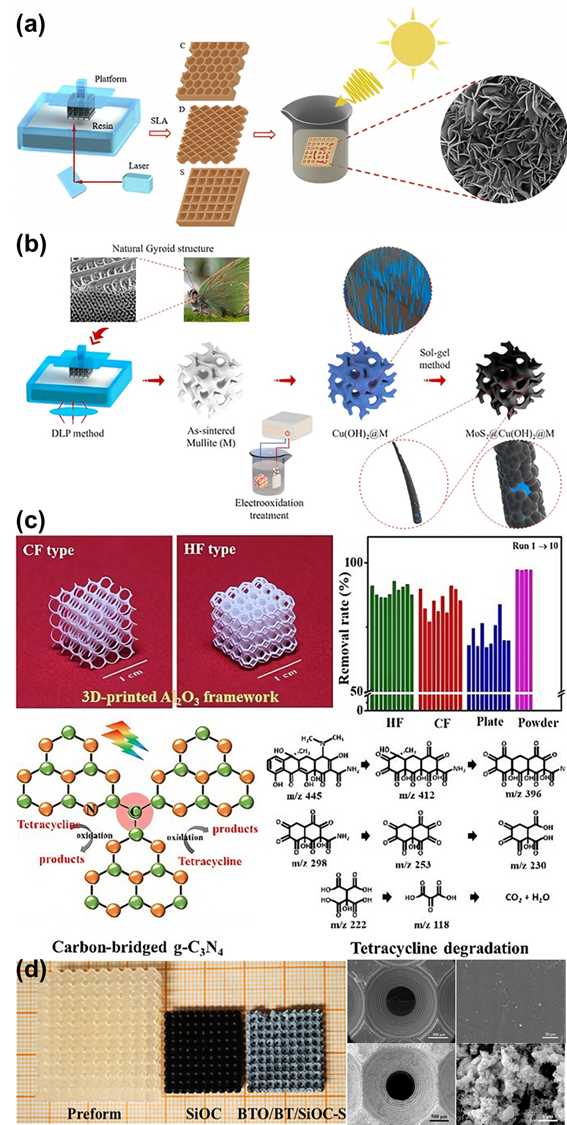
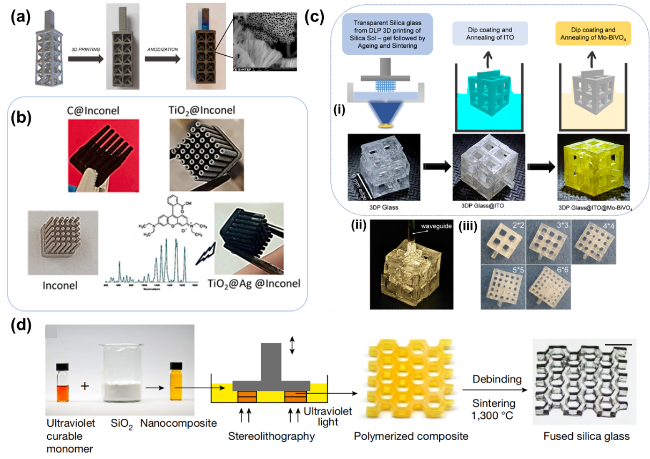
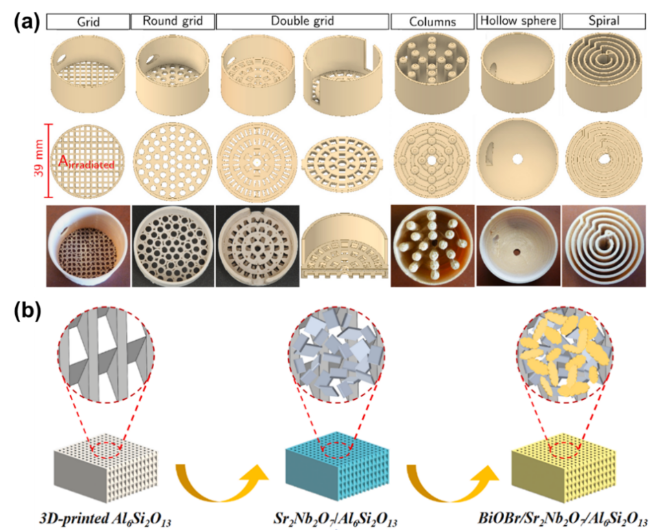
As the basis of artificial photosynthesis, the development of photocatalytic materials with appropriate and excellent catalytic performance is particularly crucial. A series of photocatalytic materials with photoresponsivity properties, including semiconductor materials, noble metal coordination compounds and organic molecular materials, have been proven to be ideal for driving photocatalytic reactions. Among these categories, semiconductor materials find extensive application in the photocatalysis field, attributed to their advantages of excellent light absorption capacity, favorable stability, abundant resource reserves, and low material costs
[99-102]. Based on their composition, semiconductor materials can currently be classified into several types of photocatalysts, including metal oxides, metal sulfides, metal oxynitrides/halide oxides and non-metals (
Fig. 4). As the earliest discovered and studied photocatalytic material within metal oxides, TiO
2 demonstrates traits of good chemical stability, non-toxicity, and low cost, thereby securing wide application in the photocatalysis field. However, traditional TiO
2 materials still have limitations in terms of their wide band gap and high recombination rate of photogenerated carriers. Therefore, a series of modification methods have been adopted for the design of materials based on the traditional TiO
2 phase. Liang et al. reviewed the research on black TiO
2, which has been widely used in environmental and energy fields due to its excellent light absorption capacity
[103]. Yang et al. systematically investigated the progress and paradigm shifts in the development of mesoporous TiO
2 photocatalysts
[104]. Mesoporous TiO
2-based structures significantly enhance the activity of photocatalysts due to their tunable pore topology, increased surface-to-volume ratio, and improved mass transfer performance. In addition to the well-known TiO
2 material, several other metal oxide, including ZnO
[105], WO
3 [106] and In
2O
3 [107], have also been widely reported to be applied in the field of photocatalysis. Nevertheless, limited by the inherent band structure of metal oxide semiconductor materials, these materials have poor absorption in the visible light range. Different from metal oxides, metal sulfide semiconductors usually have a smaller band gap, making them widely used in visible light-driven photocatalytic reactions. According to existing reports, metal sulfides, such as CdS
[108] and ZnS
[109], exhibit favorable photocatalytic effects. The application of these materials, however, has been constrained to a certain extent, hindered by issues including the inherent toxicity and low stability of metal sulfides themselves. In view of the problem that metal oxides have poor light absorption capacity due to their large band gap, researchers have proposed the design and research of metal oxynitrides
[110] and metal halide oxides
[111]. Among them, metal oxynitrides formed by doping nitrogen into metal oxides significantly expand the light absorption range to the visible light region
[112]. Taking bismuth oxyhalide, a type of metal halide oxide, as an example, its light absorption shows a red-shift from the ultraviolet to the visible light region as the contained halogen element changes from chlorine to iodine
[113]. In comparison with metal compound materials, non-metallic materials have found extensive application in the photocatalysis field, leveraging advantages of abundant resources, favorable stability, and low cost
[114-117]. Some common non-metallic materials, including graphene
[118], carbon nitride
[119] and black phosphorus
[120], have been confirmed to possess good photocatalytic activity. Wang et al. proposed loading nitrogen-doped graphene quantum dots (NGQDs) onto the surface of Mn
xCd
1-xS solid solution nanowires
[121]. The optimized Mn
0.2Cd
0.8S/NGQDs (M
0.2NG
5) composite achieved a high production rate of 6885 μmol g
-1 h
-1. Jang et al. developed a method to produce one-dimensional (1D) P-doped carbon nitride nanotubes via supramolecular self-assembly, which exhibited good photocatalytic HER activity
[122]. Shen et al. synthesized a Ni
2P-BP photocatalyst by selectively growing Ni
2P on the edges of BP nanosheets, and this catalyst achieved 100% selectivity for the photocatalytic reduction of N
2 to NH
3 [123]. In contrast to traditional photocatalysts, reticular framework materials, particularly metal-organic frameworks (MOFs) and covalent organic frameworks (COFs), have emerged as highly promising photocatalytic materials. This status is attributed to their characteristics of ultra-high surface area, customizable pore environment, and modular functionality
[124]. A large number of reports have shown that MOFs, COFs and their composite materials are excellent photocatalytic materials. Chen et al. proposed and constructed a series of covalently linked multi-component Ti-MOF/COF hybrid materials, with the optimal composite achieving a photocatalytic H
2 evolution rate of 12.8 mmol g
-1 h
-1 under simulated sunlight irradiation
[125]. Wang et al. successfully synthesized three MOFs based on tetrakis(4-carboxyphenyl)porphyrin (TCPP) via a solvothermal method, and the Zr-Ni PMOF showed a coupling reaction conversion rate of 89% for benzylamine under visible light, with a selectivity of 95% for the target product
[126]. Deng et al. designed and constructed PDI/COFs hybrid materials with different ratios via a solvothermal method, denoted as PDI/TAPB-PDA (where TAPB=1,3,5-tris(4-aminophenyl)benzene and PDA = terephthaldehyde)
[127]. Among these materials, the optimal composite PDI/TAPB-PDA-5 shows the highest photocatalytic performance for Cr(VI) reduction. To tackle issues including low solar energy utilization efficiency, high recombination rate, and limited activity, considerable research efforts have been devoted to the preparation and selection of catalysts. However, the structural impact of the catalyst substrate itself has been neglected, and traditional methods for the preparation of catalyst matrices remain restricted by factors such as the complexity of catalyst loading, as well as the stability and corrosion rate of the support.