
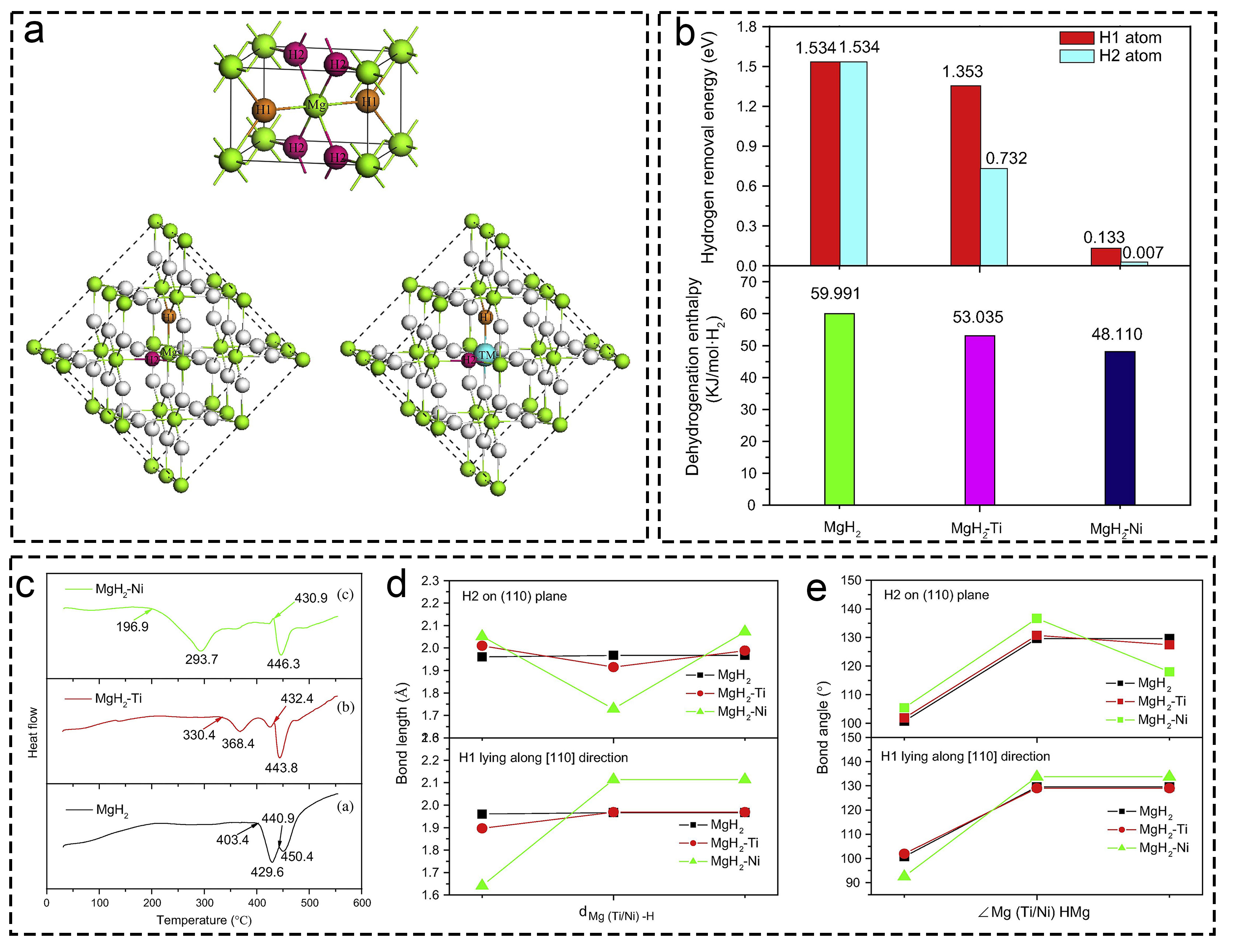
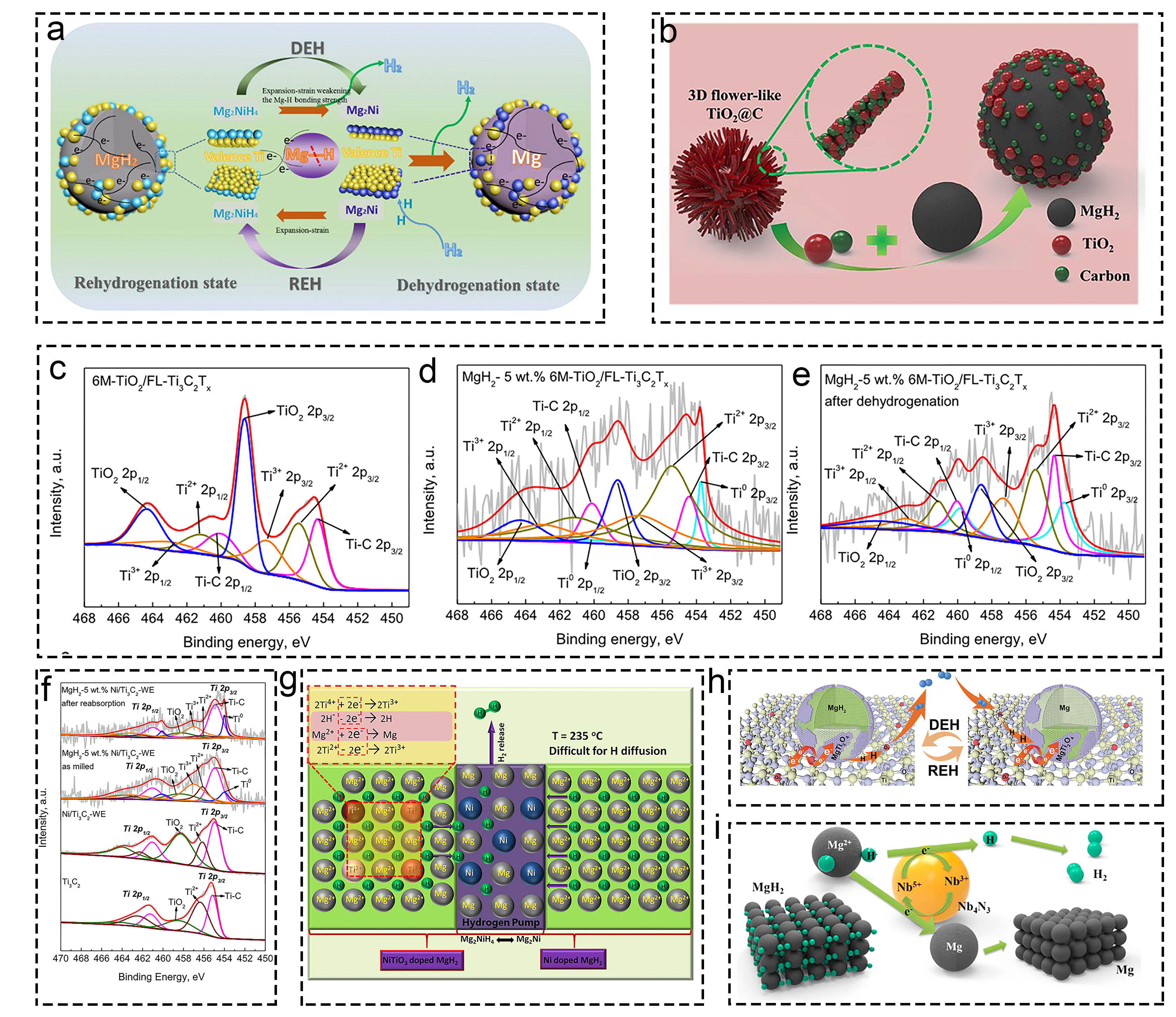
 Fig. 4 Investigations on multiple valence titanium mechanism: (a) MgH2-Ni/TiO2. Reproduced with permission [95]. Copyright 2019, Elsevier. (b) MgH2+fl-TiO2@C. Reproduced with permission [96]. Copyright 2019, Elsevier. (c-e) XPS characterization of various materials before and after dehydrogenation. Reproduced with permission [97]. Copyright 2022, Elsevier. (f) Ti3C2-based MgH2 at different states. Reproduced with permission [98]. Copyright 2021, Elsevier. (g) Ti and Ni in MgH2 hydrogen storage system. Reproduced with permission [99]. Copyright 2018, American Chemical Society. (h) MgH2/TiO2. Reproduced with permission [93]. Copyright 2022, Springer. (i) NbN doped MgH2. Reproduced with permission [100]. Copyright 2020, Elsevier.
Fig. 4 Investigations on multiple valence titanium mechanism: (a) MgH2-Ni/TiO2. Reproduced with permission [95]. Copyright 2019, Elsevier. (b) MgH2+fl-TiO2@C. Reproduced with permission [96]. Copyright 2019, Elsevier. (c-e) XPS characterization of various materials before and after dehydrogenation. Reproduced with permission [97]. Copyright 2022, Elsevier. (f) Ti3C2-based MgH2 at different states. Reproduced with permission [98]. Copyright 2021, Elsevier. (g) Ti and Ni in MgH2 hydrogen storage system. Reproduced with permission [99]. Copyright 2018, American Chemical Society. (h) MgH2/TiO2. Reproduced with permission [93]. Copyright 2022, Springer. (i) NbN doped MgH2. Reproduced with permission [100]. Copyright 2020, Elsevier.
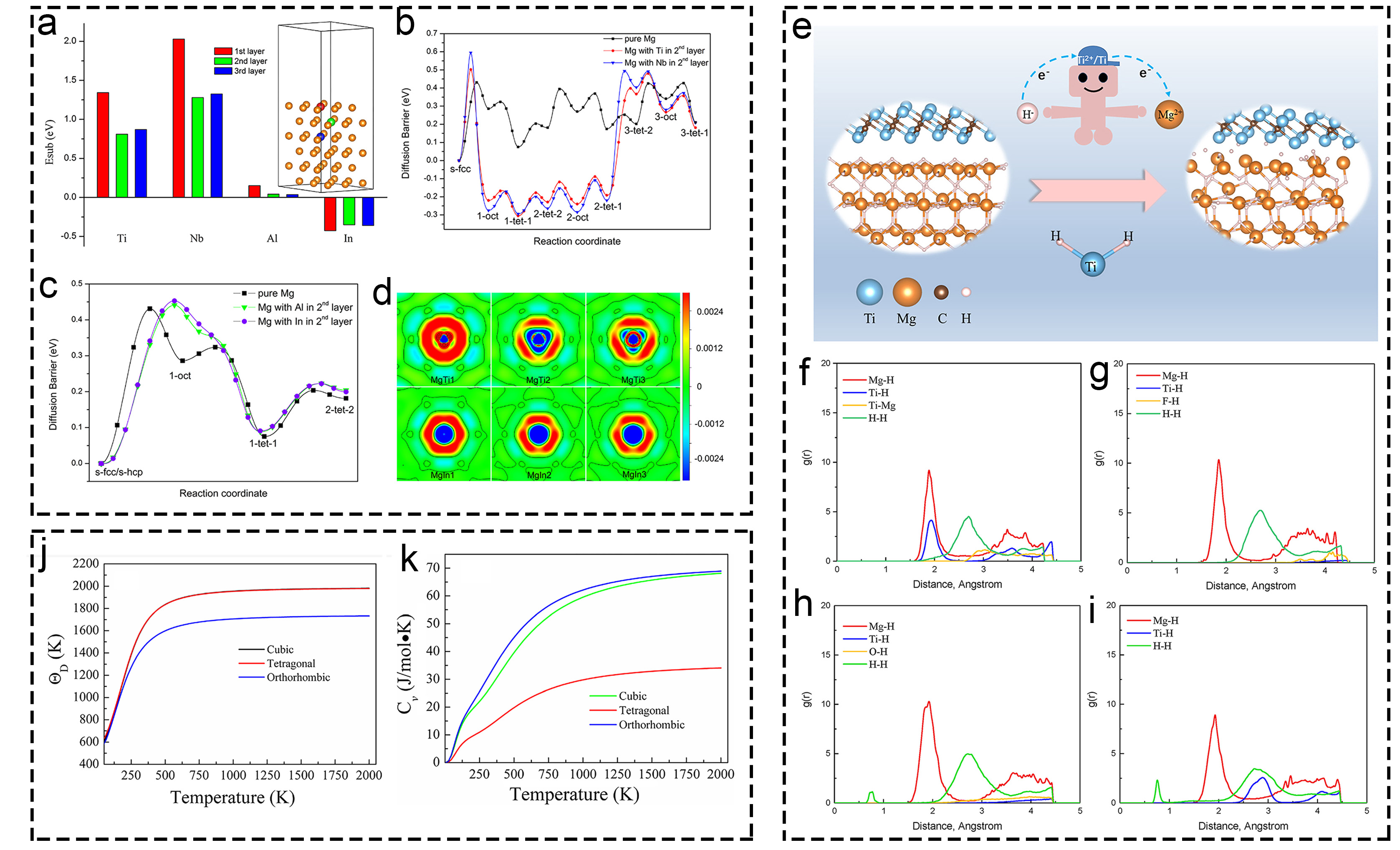 Fig. 6 Theoretical research of Ti-based additives on hydrogen storage based on pure DFT calculation. (a-d) The energy changes of Ti, Nb, Al, and In. Reproduced with permission [109]. Copyright 2017, Elsevier. (e-i) Mg-H, Ti-H and Ti-Mg bond of various structures. Reproduced with permission [111]. Copyright 2021, Elsevier. (j-k) Thermodynamic properties of TiH2 in Debye temperature and heat capacity. Reproduced with permission [112]. Copyright 2020, Wiley.
Fig. 6 Theoretical research of Ti-based additives on hydrogen storage based on pure DFT calculation. (a-d) The energy changes of Ti, Nb, Al, and In. Reproduced with permission [109]. Copyright 2017, Elsevier. (e-i) Mg-H, Ti-H and Ti-Mg bond of various structures. Reproduced with permission [111]. Copyright 2021, Elsevier. (j-k) Thermodynamic properties of TiH2 in Debye temperature and heat capacity. Reproduced with permission [112]. Copyright 2020, Wiley.
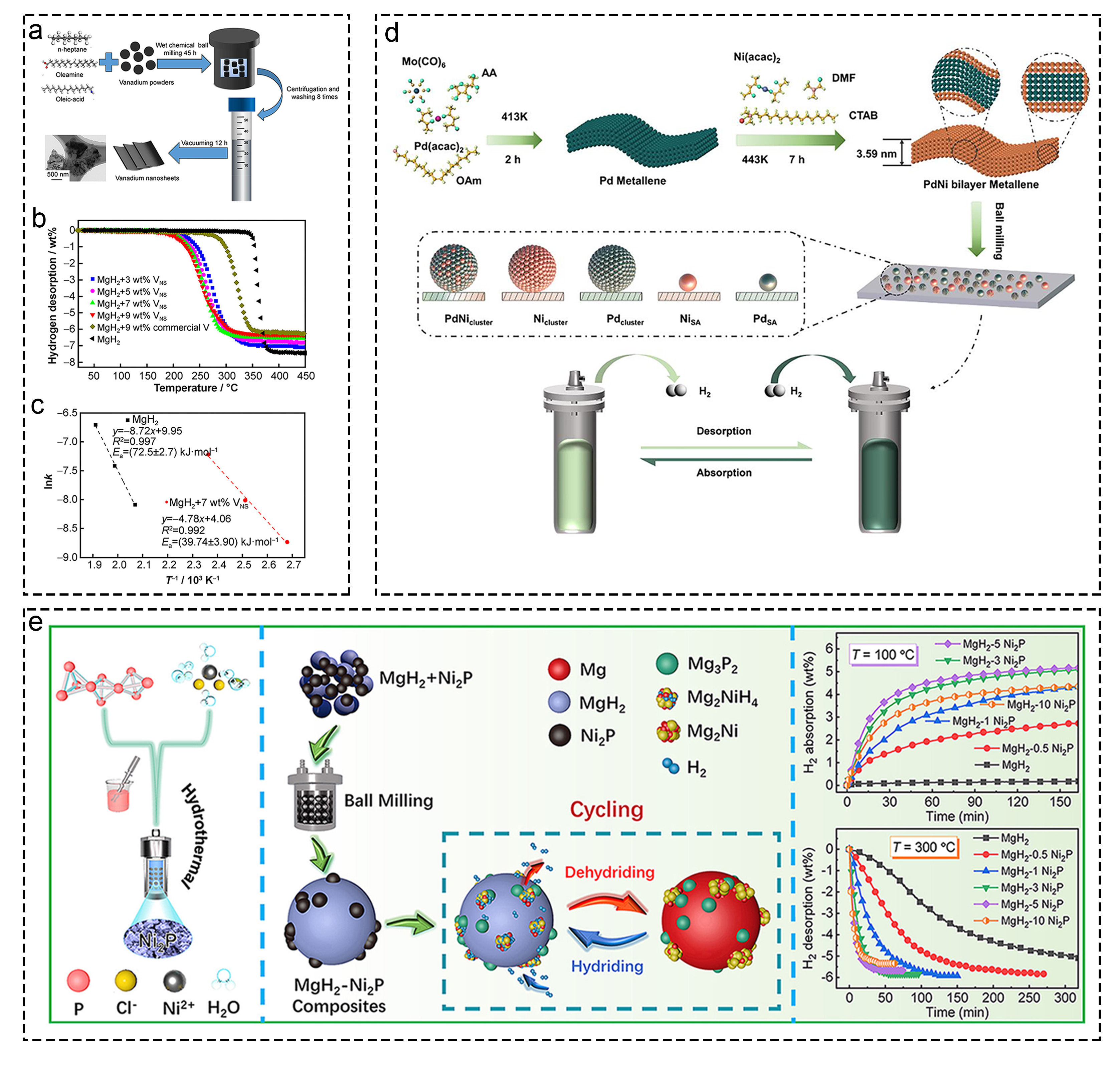 Fig. 8 Corresponding information and presentation of various catalysts on hydrogen storage of MgH2 and MgH2-based composites. (a-c) Vanadium nanosheets. Reproduced with permission [137]. Copyright 2021, Nonferrous Metals Society of China. (d) MgH2-PdNi, Reproduced with permission [138]. Copyright 2023, Wiley. (e) MgH2-Ni2P. Reproduced with permission [140]. Copyright 2022, Elsevier.
Fig. 8 Corresponding information and presentation of various catalysts on hydrogen storage of MgH2 and MgH2-based composites. (a-c) Vanadium nanosheets. Reproduced with permission [137]. Copyright 2021, Nonferrous Metals Society of China. (d) MgH2-PdNi, Reproduced with permission [138]. Copyright 2023, Wiley. (e) MgH2-Ni2P. Reproduced with permission [140]. Copyright 2022, Elsevier.
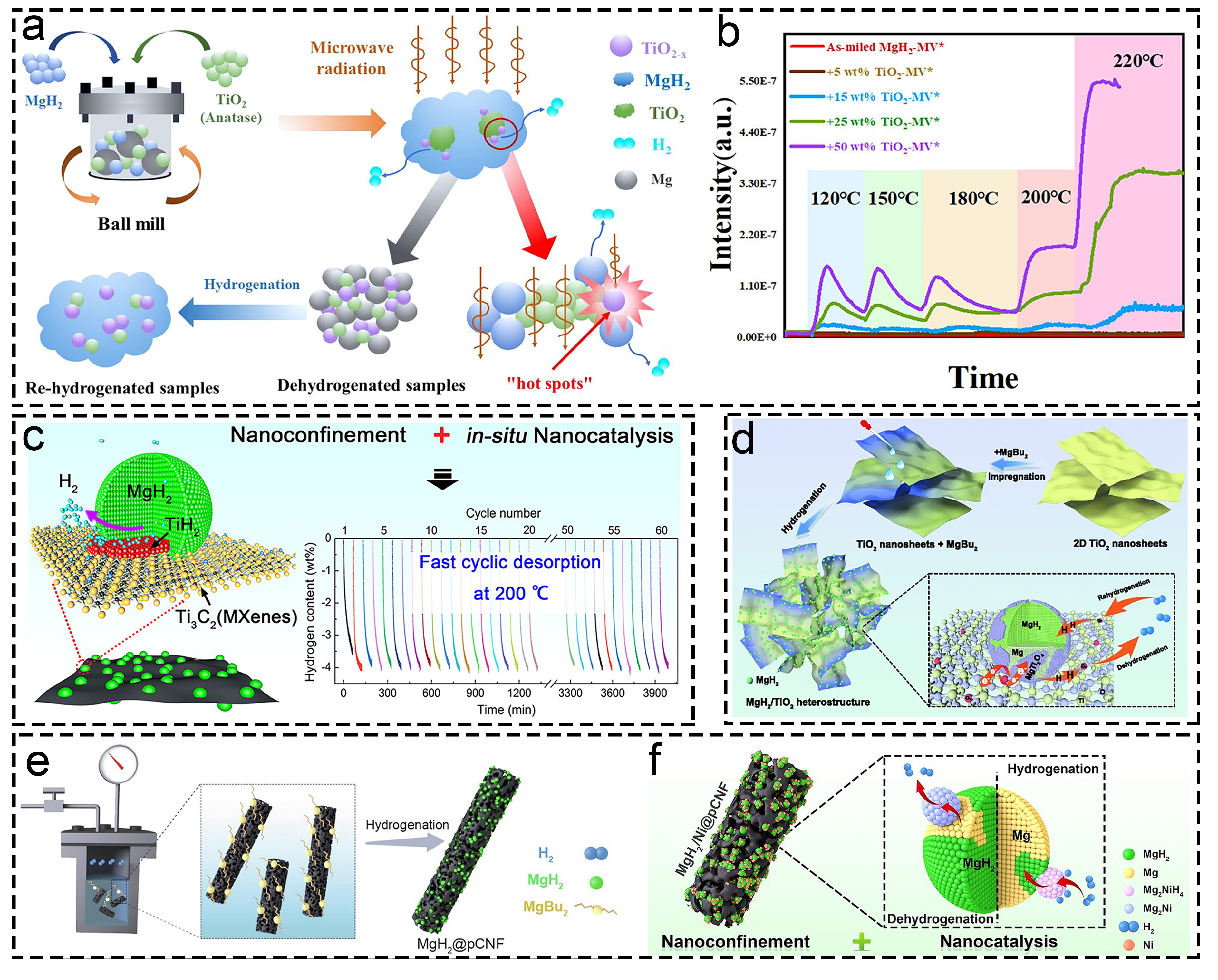 Fig. 11 Corresponding information and presentation of different catalysts on hydrogen storage of MgH2 and MgH2-based composites. (a, b) MgH2-TiO2. Reproduced with permission [190]. Copyright 2022, Elsevier. (c) MgH2@Ti-MX. Reproduced with permission [40]. Copyright 2021, American Chemical Society. (d) MgH2/TiO2. Reproduced with permission [93]. Copyright 2022, Springer. (e, f) MgH2@pCNF. Reproduced with permission [192]. Copyright 2022, Elsevier.
Fig. 11 Corresponding information and presentation of different catalysts on hydrogen storage of MgH2 and MgH2-based composites. (a, b) MgH2-TiO2. Reproduced with permission [190]. Copyright 2022, Elsevier. (c) MgH2@Ti-MX. Reproduced with permission [40]. Copyright 2021, American Chemical Society. (d) MgH2/TiO2. Reproduced with permission [93]. Copyright 2022, Springer. (e, f) MgH2@pCNF. Reproduced with permission [192]. Copyright 2022, Elsevier.
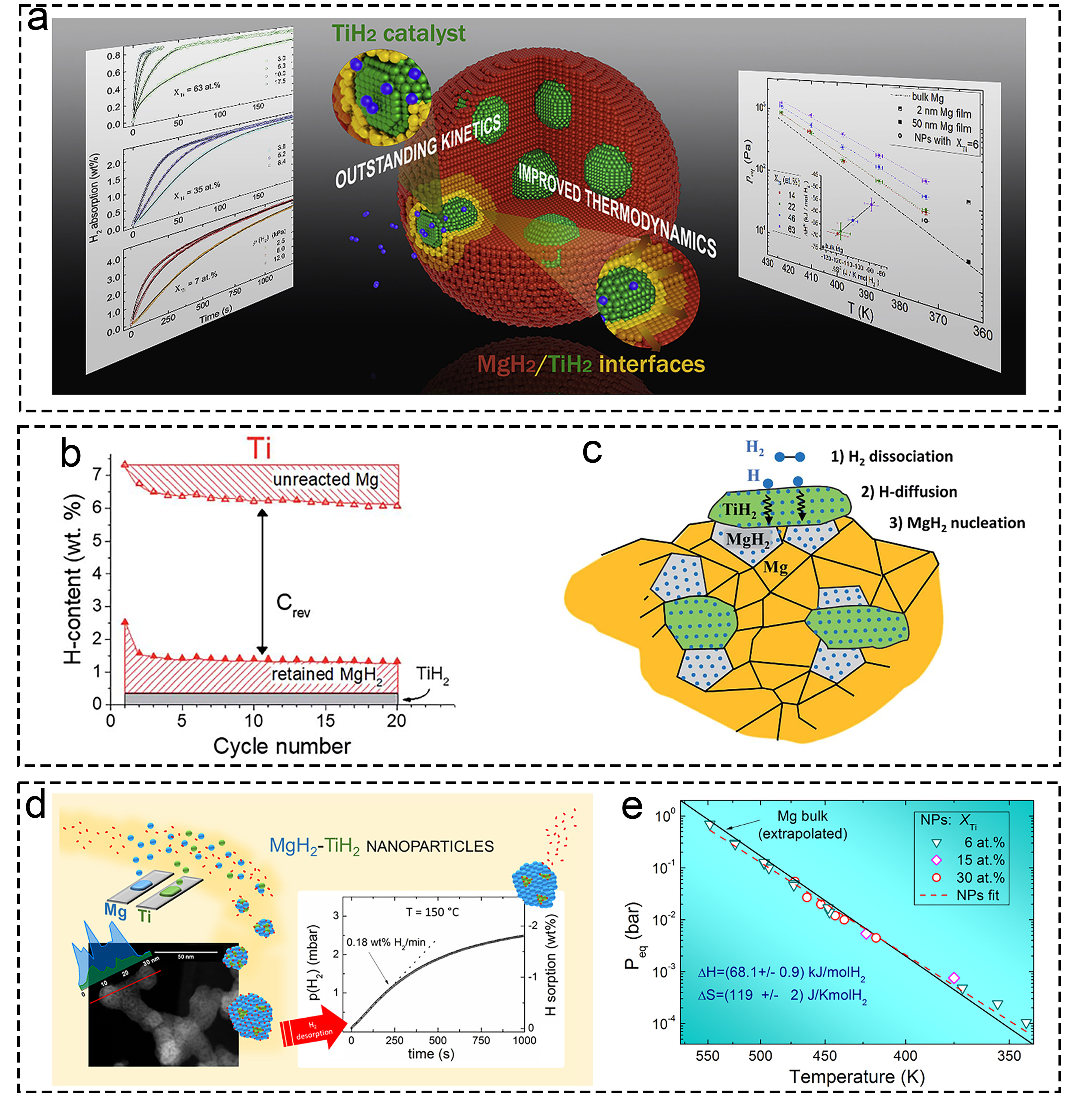 Fig. 12 Corresponding characterization from the morphology, performance and mechanism characterization of Ti-based materials. (a) Reproduced with permission [249]. Copyright 2020, Elsevier. (b, c) Reproduced with permission [251]. Copyright 2019, Royal Society of Chemistry. (d, e) 3D Ti3C2 MXene. Reproduced with permission [252]. Copyright 2017, American Chemical Society.
Fig. 12 Corresponding characterization from the morphology, performance and mechanism characterization of Ti-based materials. (a) Reproduced with permission [249]. Copyright 2020, Elsevier. (b, c) Reproduced with permission [251]. Copyright 2019, Royal Society of Chemistry. (d, e) 3D Ti3C2 MXene. Reproduced with permission [252]. Copyright 2017, American Chemical Society.
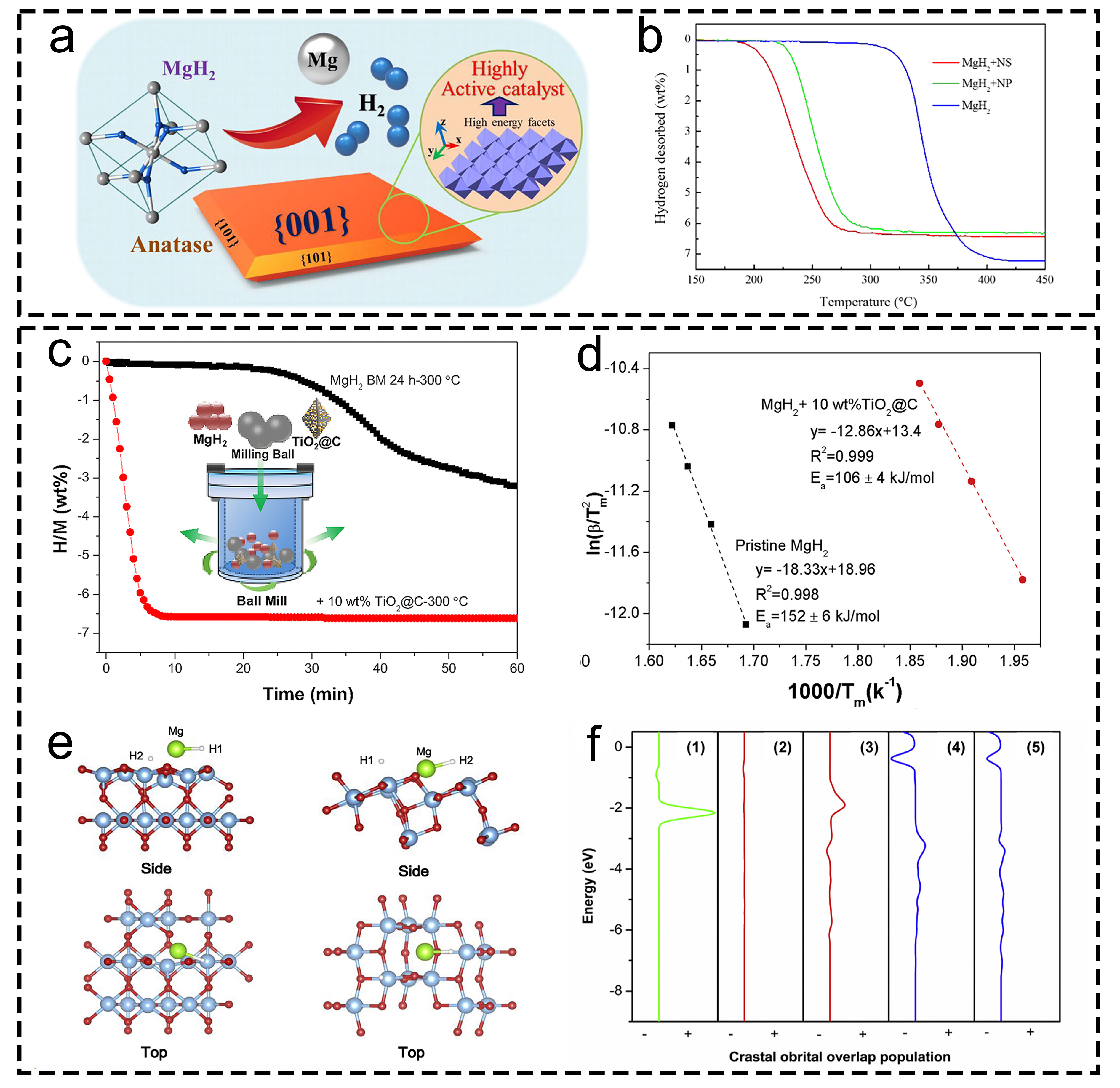 Fig. 13 Corresponding information and presentation of different catalysts on hydrogen storage of MgH2 and MgH2-based composites. (a, b) TiO2 NS with exposed {001} facets. Reproduced with permission [72]. Copyright 2019, Royal Society of Chemistry. (c-f) TiO2@C. Reproduced with permission [274]. Copyright 2018, Elsevier.
Fig. 13 Corresponding information and presentation of different catalysts on hydrogen storage of MgH2 and MgH2-based composites. (a, b) TiO2 NS with exposed {001} facets. Reproduced with permission [72]. Copyright 2019, Royal Society of Chemistry. (c-f) TiO2@C. Reproduced with permission [274]. Copyright 2018, Elsevier.
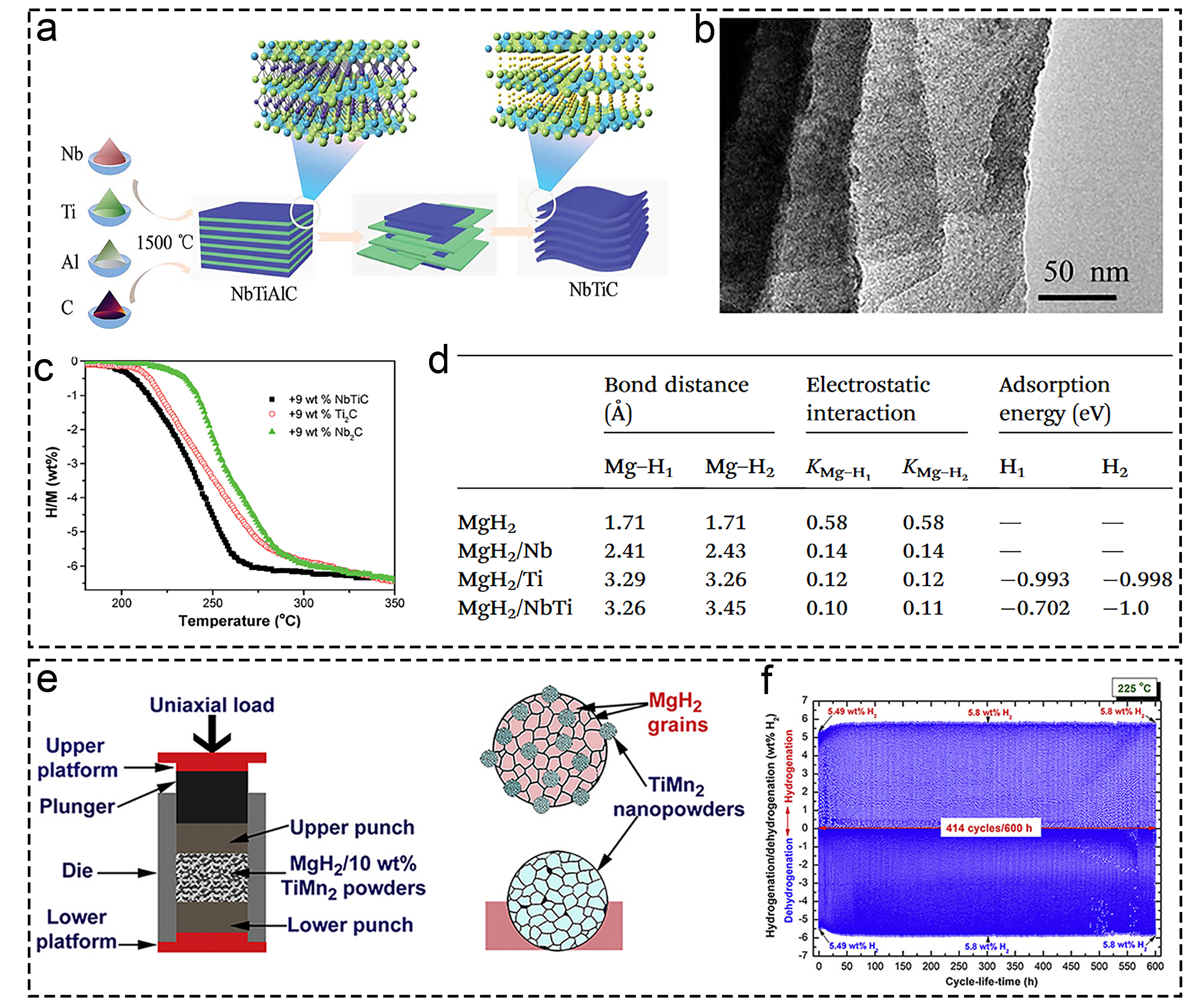 Fig. 15 The corresponding characterization from the morphology, performance and mechanism characterization of polymetallic titanium compounds. (a-d) NbTi cluster. Reproduced with permission [299]. Copyright 2019, Royal Society of Chemistry. (e, f) TiMn2 compound. Reproduced with permission [300]. Copyright 2019, Royal Society of Chemistry.
Fig. 15 The corresponding characterization from the morphology, performance and mechanism characterization of polymetallic titanium compounds. (a-d) NbTi cluster. Reproduced with permission [299]. Copyright 2019, Royal Society of Chemistry. (e, f) TiMn2 compound. Reproduced with permission [300]. Copyright 2019, Royal Society of Chemistry.
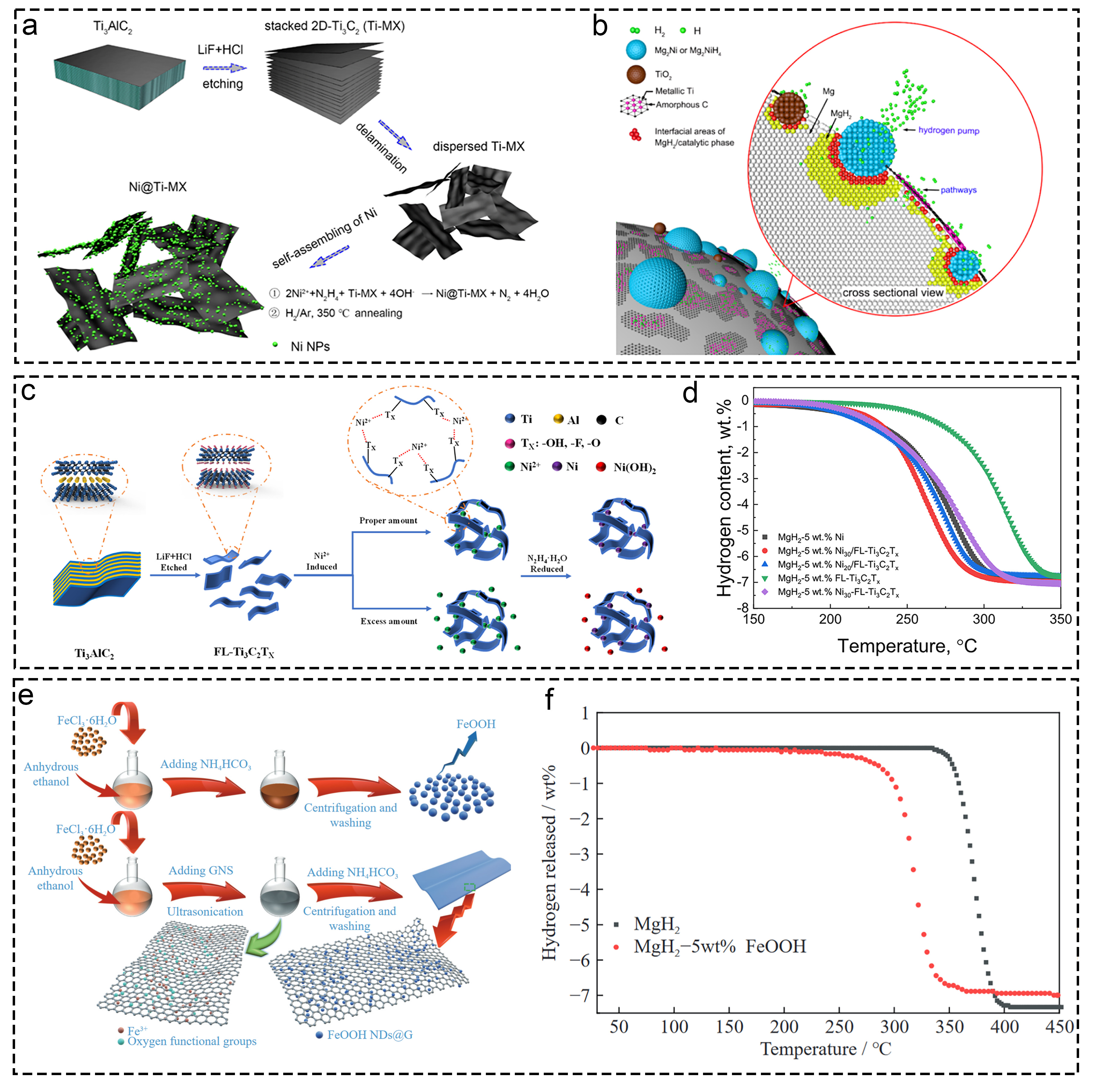 Fig. 16 The corresponding characterization from the morphology, performance and mechanism characterization of materials supported on Ti-based supports. (a, b) Ti3C2 MXene-based catalyst (Ni@Ti-MX). Reproduced with permission [315]. Copyright 2020, American Chemical Society. (c, d) few-layer Ti3C2Tx (FL-Ti3C2Tx) supporting highly dispersed nano-Ni particles. Reproduced with permission [316]. Copyright 2020, American Chemical Society. (e, f) graphene nanosheet-supported FeOOH nanodots. Reproduced with permission [318]. Copyright 2022, Springer.
Fig. 16 The corresponding characterization from the morphology, performance and mechanism characterization of materials supported on Ti-based supports. (a, b) Ti3C2 MXene-based catalyst (Ni@Ti-MX). Reproduced with permission [315]. Copyright 2020, American Chemical Society. (c, d) few-layer Ti3C2Tx (FL-Ti3C2Tx) supporting highly dispersed nano-Ni particles. Reproduced with permission [316]. Copyright 2020, American Chemical Society. (e, f) graphene nanosheet-supported FeOOH nanodots. Reproduced with permission [318]. Copyright 2022, Springer.
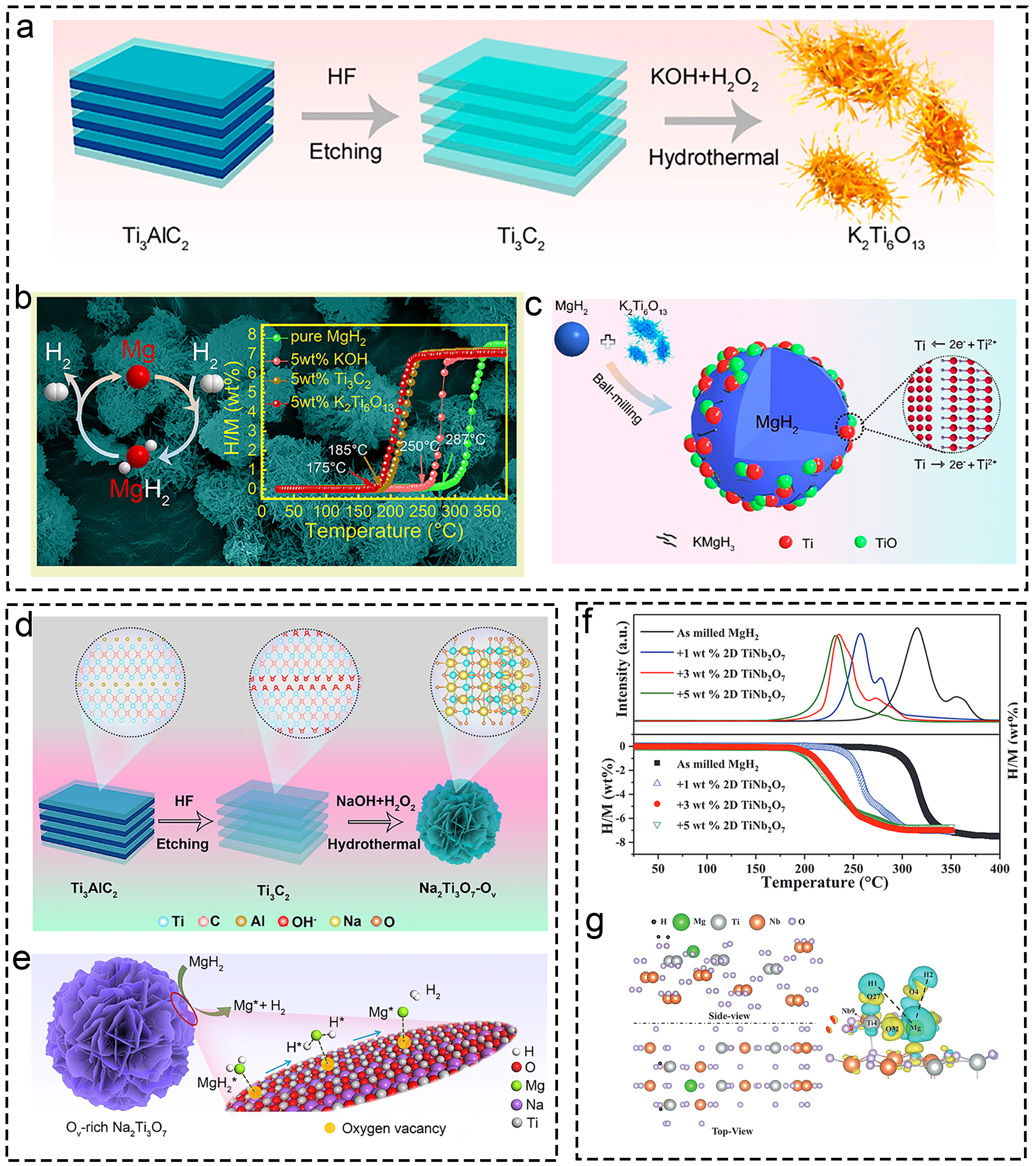 Fig. 17 Corresponding characterization from the morphology, performance and mechanism characterization of Ti-based materials. (a-c) K2Ti6O13. Reproduced with permission [333]. Copyright 2020, American Chemical Society. (d, e) Oxygen Vacancies modified Na2Ti3O7. Reproduced with permission [90]. Copyright 2022, American Chemical Society. (f, g) TiNb2O7. Reproduced with permission [335]. Copyright 2022, Wiley.
Fig. 17 Corresponding characterization from the morphology, performance and mechanism characterization of Ti-based materials. (a-c) K2Ti6O13. Reproduced with permission [333]. Copyright 2020, American Chemical Society. (d, e) Oxygen Vacancies modified Na2Ti3O7. Reproduced with permission [90]. Copyright 2022, American Chemical Society. (f, g) TiNb2O7. Reproduced with permission [335]. Copyright 2022, Wiley.